Use data from Figure 20-12 to determine the temperature at which t 1/2 for the first-order decomposition
Question:
Use data from Figure 20-12 to determine the temperature at which t1/2 for the first-order decomposition of N2O5 in CCl4 is 2.00 h.
Figure 20-12
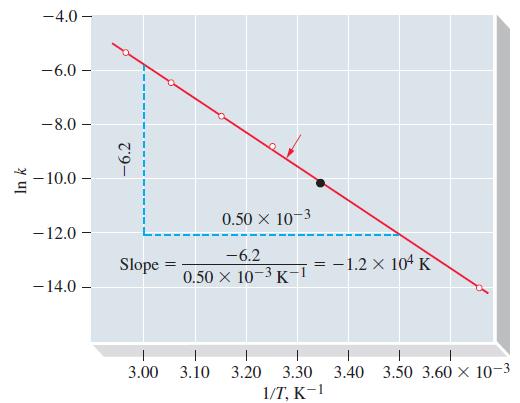
Transcribed Image Text:
In k -4.0 - -6.0- -8.0 -10.0- -12.0 - - 14.0 -6.2 Slope = 3.00 0.50 × 10-3 -6.2 0.50 x 10-3K-1 3.10 3.20 3.30 1/T, K-1 -1.2 x 104 K 3.40 3.50 3.60 × 10-3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Analyze First find the rate constant k corresponding to a 200 h halflife This can be done by using t...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) What is the half-life of the first-order decomposition of N 2 O 5 at 75.0 C? Use data from Example 20-9. (B) At what temperature will it take 1.50 h for two-thirds of a sample of N 2 O 5 in CCl 4...
-
In Example 13-12, we used the vant Hoff equation to determine the temperature at which for the reaction 2 SO 2 (g) + O 2 (g) 2 SO 3 (g). Obtain another estimate of this temperature with data from...
-
The 50-mm-diameter cylinder is made from Am 1004-T61 magnesium and is placed in the clamp when the temperature is T 1 = 15°C. If the two 304-stainless-steel carriage bolts of the clamp each have...
-
Ha: μ1 > μ2 The null hypothesis is H0: μ1 = μ2 and the alternative hypothesis is as specified. We have provided data from a simple random...
-
Pockey Company has assembled the following market data, based on commercial real estate sales in the area in the past six months, which can be used to indirectly estimate the selling price of a...
-
The following transactions occurred in June at Fast Wheels, Inc., a custom bicycle manufacturer: 1. Purchased $20,000 of materials. 2. Issued $1,000 of supplies from the materials inventory. 3....
-
Following are observed frequencies. The null hypothesis is H0: p1 = 0.4, p2 = 0.25, p3 = 0.05, p4 = 0.1, p5 = 0.2. a. Compute the expected frequencies. b. Compute the value of X2. c. How many degrees...
-
Jose Pena and Joseph Antenucci were medical doctors who were partners in a medical practice. Both doctors treated Elaine Zuckerman during her pregnancy. Her son, Daniel Zuckerman, was born with...
-
Tick the true statements relating to implementing costing strategies: Businesses will assess any action based on the costs involved and therefore a financial assessment is necessary for any...
-
What is the approximate concentration of A remaining after 110 s in the (a) Zero-order, (b) First-order, (c) Second-order reaction? I Time, S 0 25 50 75 100 150 200 250 [A], M 1.00 0.78 0.61 0.47...
-
Use data from Table 20.3 to establish the order of reaction (20.7) with respect to HgCl 2 and C 2 O 4 2- and also the overall order of the reaction. Table 20.3 Reaction (20.7) TABLE 20.3 Kinetic Data...
-
Referring to Fig. 6.25, sketch the intermediate steps you would recommend in the forging of a wrench.
-
Viva sells its waterproof phone case for $90 per unit. Fixed costs total $194,400, and variable costs are $36 per unit. (1) Determine the contribution margin per unit. Contribution margin (2)...
-
Grace Inc. owned all of Mary Co. Although the Investment in Mary Co. account had a balance of $862,000, the subsidiary's 12,000 shares had an underlying book value of only $55 per share. On January...
-
where q is the reduction in emissions (in pounds of pollutant per day) and C is the daily cost to the firm (in dollars) of this reduction. The government will pay subsidies in the amount of $550 per...
-
The plant manager of Orlando Electronics Company is considering the purchase of new automated assembly equipment. The new equipment will cost $189,000. The manager believes that the new investment...
-
How do historical legacies of colonization, imperialism, and systemic oppression manifest in contemporary structures of power and privilege, and what strategies can be employed to dismantle these...
-
The Alaska Airlines balance sheet dated December 31, 2011, included the following ($ in millions): Property and equipment Aircraft and other flight equipment ........$4,041.8 Other property and...
-
Show that the peak of the black body spectrum as a function of ? is given by eq. (22.14) kg T Wmax = 2.82
-
The trial balance for Pioneer Advertising Agency is shown in Illustration 3-3, p. 100. In lieu of the adjusting entries shown in the text at October 31, assume the following adjustment data. 1....
-
The income statement of Benning Co. for the month of July shows net income of $1,400 based on Service Revenue $5,500, Wages Expense $2,300, Supplies Expense $1,200, and Utilities Expense $600. In...
-
A partial adjusted trial balance of Sila Company at January 31, 2010, shows the following. InstructionsAnswer the following questions, assuming the year begins January 1.(a) If the amount in Supplies...
-
Administrative agencies at various levels of government can work together and share the responsibility of creating and enforcing regulations.Explain
-
Marketing communicators aim to build customer-brand relationships and guide customers through the five As of the customer journey. What is the first of the five As?
-
The Commissioner revokes an intermediary's license because the agent ignored an order of restraint. How much time must pass before the intermediary can reapply for a license?

Study smarter with the SolutionInn App


