Use data from Figure 9-9 to predict the type of cubic unit cell adopted by (a) BaO;
Question:
Use data from Figure 9-9 to predict the type of cubic unit cell adopted by
(a) BaO;
(b) CuI;
(c) LiS2.
(The radii of Ba2+ and S2- ions are 135 and 198 pm, respectively.)
Figure 9-9
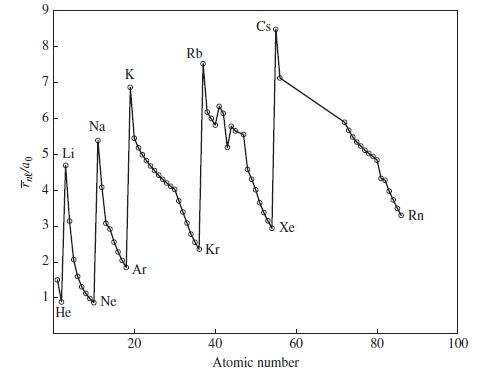
Transcribed Image Text:
Tne/ao 9 8 7 6 5 Li 3 2 1 He Na Ne K Ar 20 Rb Kr Csa Xe 40 60 Atomic number 80 Rn 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
To predict the type of cubic unit cell adopted by a compound we can use the following guidelines Sim...View the full answer

Answered By

Sagar Kumar
I am Mechanical Engineer with CGPA of 3.98 out of 4.00 from Pakistan. I went to Government Boys Degree College, Sehwan for high school studies.
I appeared in NUST Entrance Exam for admission in university and ranked #516. My mathematics are excellent and I have participated in many math competitions and also won many of them. Recently, I participated in International Youth Math Challenge and was awarded with Gold Honor. Now, I am also an ambassador at International Youth Math Challenge,
I have been teaching when I was in 9th class class year 2012. I have taught students from 6th class to university level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use data from Figure 9-11 to predict the type of cubic unit cell adopted by (a) CaO; (b) CuCl; (c) LiO 2 (the radius of the O 2 - ion is 128 pm). Figure 9-11 155 59 Li+ Be+ 102 K Na Mg 190 160 Na...
-
The table below lists the ionic radii for the cations and anions in three different ionic compounds. Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of...
-
The table below lists the ionic radii for the cations and anions in three different ionic compounds. Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of...
-
Assume that the average talk time on an Apple iPhone is 20 hours and that this time follows the exponential probability distribution. What is the probability that a randomly selected iPhone will...
-
A farmer made a contract in April to sell to a grain dealer forty thousand bushels of corn to be delivered in October. On June 3, the farmer unequivocally informed the grain dealer that he was not...
-
Two radioactive materials that alpha decay, 238U and 232Th, and one that beta decays, 40K, are sufficiently abundant in granite to contribute significantly to the heating of Earth through the decay...
-
Refer to Samsungs financial statements in Appendix A. Compute its debt ratio as of December 31, 2015, and December 31, 2014. Data From Samsung Financial Statement Appendix A Samsung Electronics Co.,...
-
Allowance Method for Accounting for Bad Debts At the beginning of 2010, EZ Tech Companys accounts receivable balance was $140,000 and the balance in Allowance for Doubtful Accounts was $2,350. EZ...
-
A child bounces a 48 g superball on the side- walk. The velocity change of the superball is from 23 m/s downward to 13 m/s upward. If the contact time with the sidewalk is 1 800 s, what is the...
-
Potassium chloride has the same crystal structure as NaCl. Careful measurement of the internuclear distance between K + and Cl - ions gave a value of 314.54 pm. The density of KCl is 1.9893 g/cm 3 ....
-
Two views, a top and side view, for the unit cell for rutile (TiO 2 ) are shown here. (a) How many titanium atoms (blue) are in this unit cell? (b) How many oxygen atoms (red) are in this unit cell?
-
Terminology, Relationships, Computations, Entries Instructions Complete the following statements by filling in the blanks. (a) In a period in which a taxable temporary difference reverses, the...
-
What categories of services should Medicare cover? Should its coverage be identical to employment related benefit packages or should it vary in some respects? What items might be more, or less,...
-
Research indicates that how people rate whether government is working well is based largely on what factor?
-
How you feel about the job market you will be participating in. What does this mean for you as a job seeker in this market?
-
The VA lottery group has read your report and provided you with a new assignment: describe a sales pitch in which you describe the main drivers of lottery sales, and in particular address the...
-
a) What are green business opportunities, explain in the context of Environmental Threat and Opportunity Profile (ETop b) What are the responsibilities entrepreneurs have to suppliers, customers and...
-
On January 1, 2013, VKI Corporation awarded 12 million of its $1 par common shares to key personnel, subject to forfeiture if employment is terminated within three years. On the grant date, the...
-
Information graphics, also called infographics, are wildly popular, especially in online environments. Why do you think infographics continue to receive so much attention? How could infographics be...
-
Calculating Annuity Values your company will generate $65,000 in annual revenue each year for the next eight years from a new information database, if the appropriate interest rate is 8.5 percent,...
-
Calculating Annuity Values If you deposit $3,000 at the end of each of the next 20 years into an account paying 10.5 percent interest, how much money will you have in the account in 20 years how much...
-
Calculating Annuity Values you want to have $80,000 in your savings account 10 years from now, and youre prepared to make equal annual deposits into the account at the end of each year, if the...
-
Assuming that the directors decide to declare total dividends in the amount of $393,900, determine how much each class of shares should receive if the preferred shares are non-cumulative and...
-
During the year, the corporation completed a number of transactions affecting the stockholders' equity. They are summarized as follows: a. Issued 521,500 shares of common stock at $9, receiving cash....
-
A company has to choose one of the following two mutually exclusive projects. Investments required for each project is Rs. 15,000. Both the projects have to be depreciated on straight- line basis....

Study smarter with the SolutionInn App


