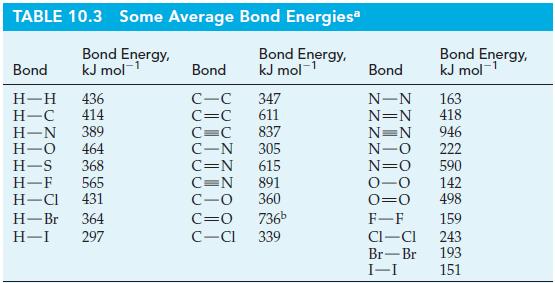
Use data from Table 10.3 to estimate the enthalpy change ( r H) for the following reaction.
Question:
Use data from Table 10.3 to estimate the enthalpy change (ΔrH) for the following reaction.
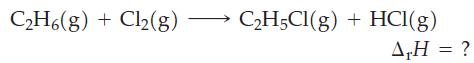 Table 10.3
Table 10.3
Transcribed Image Text:
C₂H6(g) + Cl2(g) C₂H5Cl(g) + HCl(g) AH = ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
C2H6 Cl2g CHC HClg AH ...View the full answer

Answered By

Antony Sang
I am a research and academic writer whose work is outstanding. I always have my customer's interests at heart. Time is an important factor in our day to day life so I am always time conscious. Plagiarism has never been my thing whatsoever. I give best Research Papers, Computer science and IT papers, Lab reports, Law, programming, Term papers, English and literature, History, Math, Accounting, Business Studies, Finance, Economics, Business Management, Chemistry, Biology, Physics, Anthropology, Sociology, Psychology, Nutrition, Creative Writing, Health Care, Nursing, and Articles.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use Table 8.4 to estimate the enthalpy change for each of the following reactions: a. H2C == O (g) + HCl (g) H3C - O - Cl (g) b. H2O2 (g) + 2CO (g) H2 (g) + CO2 (g) (c). 3H2C == CH2 (g) C6H12 (g)...
-
Ammonia is produced directly from nitrogen and hydrogen by using the Haber process. The chemical reaction is N2 (g) + 3H2 (g) 2NH3 (g) (a) Use Table 8.4 to estimate the enthalpy change for the...
-
(a) Use bond enthalpies to estimate the enthalpy change for the reaction of hydrogen with ethylene: H2 (g) + C2H4 (g) C2H6 (g) (b) Calculate the standard enthalpy change for this reaction, using...
-
Education is a very important job because it can change and shape people's lives. It gives people the knowledge, skills, and attitudes they need to be successful in their personal and work lives. As...
-
The Snyder Mfg. Co., being a large user of coal, entered into separate contracts with several coal companies. In each contract it was agreed that the coal company would supply coal during the entire...
-
Indole reacts with electrophile at C3 rather than at C2. Draw resonance forms of the intermediate cations resulting from reaction at C2 and C3, and explain the observed results.
-
On December 14, 2011, appellant Aaron Olson contracted to receive telephone service from respondent CenturyLink and also applied for reduced-rate service that CenturyLink provides through Minnesotas...
-
Stacy Corporation had income before income taxes for 2012 of $6,300,000. In addition, it suffered an unusual and infrequent pretax loss of $770,000 from a volcano eruption. The corporations tax rate...
-
Find the perimeter of the figure shown. Express the perimeter using the same unit of measure that appears on the given sides. 13 yd Perimeter = 5 yd 5 yd 13 yd 18 yd 18 yd
-
One of the chemical reactions that occurs in the formation of photochemical smog is O 3 + NO NO 2 + O 2 . Estimate r H for this reaction by using appropriate Lewis structures and data from Table...
-
A reaction involved in the formation of ozone in the upper atmosphere is O 2 2 O. Without referring to Table 10.3, indicate whether this reaction is endothermic or exothermic. Explain. Table 10.3...
-
The unadjusted trail balance Garden Designs at its month end, April 30, 2014, is as follows: Additional information: 1. $500 of the unearned revenue has been earned by April 30, 2014. 2. On April 1,...
-
What are the internal and external stressors you have experienced or continue to experience? What motivates you to enter your classroom every day? How will you influence the stakeholders in your...
-
There are 5 Sequential steps to Effective Self Leadership List and Describe the 5 Sequential Steps. Part 2 (6 Marks) Setting Goals and objectives are a critical Start. Write on Goal that you can...
-
What are the challenges of today's educational climate? What does your learning environment look like? Who are your learners? How will you evolve to become a teacher- leader in today's educational...
-
What are some effective marketing channels for reaching customers?
-
Differentiate between benign and malignant tumors? Which one is lethal and why?
-
The Kwok Companys inventory balance on December 31, 2013, was $165,000 (based on a 12/31/13 physical count) before considering the following transactions: 1. Goods shipped to Kwok f.o.b. destination...
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
Norman Gerrymander has just received a $2 million bequest. How should he invest it? There are four immediate alternatives. a.Investment in one-year U.S. government securities yielding 5 percent. b. A...
-
Show that your answers to Practice Question 7 are consistent with the rate of return rule for investment decisions. a.Investment in one-year U.S. government securities yielding 5 percent. b. A loan...
-
Take another look at investment opportunity (d) in Practice Question 7. Suppose a bank offers Norman a $600,000 personal loan at 8 percent. (Norman is a long-time customer of the bank and has an...
-
As a project manager, how can you optimize and streamline project communications?
-
Make a program in c language Question 02 (18) Vital signs are measurements of the body's most basic functions. The four main vital signs routinely monitored by medical professionals and health care...
-
Summarize the article titled "Supply chain integration and novelty-centered business model design: An organizational learning perspective" Authors: Taiwen Feng, Shan Yang, Hongyan Sheng. And also...

Study smarter with the SolutionInn App


