A reaction involved in the formation of ozone in the upper atmosphere is O 2 2
Question:
A reaction involved in the formation of ozone in the upper atmosphere is O2 → 2 O. Without referring to Table 10.3, indicate whether this reaction is endothermic or exothermic. Explain.
Table 10.3
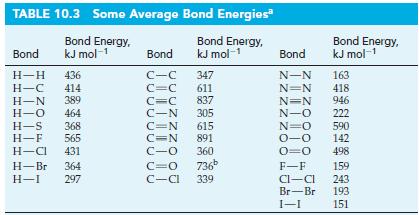
Transcribed Image Text:
TABLE 10.3 Some Average Bond Energies Bond Energy, kJ mol-1 Bond Energy, kJ mol-1 Bond H-H 436 H-C 414 H-N 389 H-O 464 H-S 368 H-F 565 H-Cl 431 H-Br 364 H-I 297 Bond C-C C=C 347 611 837 C=C C-N 305 C=N 615 C=N 891 360 C-O C=O 736⁰ C-Cl 339 Bond Energy, kJ mol-1 Bond N-N 163 N=N 418 N=N 946 N-O 222 N=O 590 0-0 142 0=0 498 F-F Cl-Cl Br-Br 1-1 159 243 193 151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine whether the reaction O2 2 O is endothermic or exotherm...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A reaction involved in the metabolism of sugars is the splitting of fructose-1,6-diphosphate to give glyceraldehyde-3-phosphate and dihydroxyacetone phosphate. In the living system, this retro-aldol...
-
Write sentences for the long-term direction and strategic path that management intends to follow. "Where we are headed?" and should explain why the direction in which you intend to point the company...
-
Explain what energy terms are involved in the formation of an ionic solid from atoms. In what way should these terms change (become larger or smaller) to give the lowest energy possible for the solid?
-
An important U.S. government organization charged with setting human resource management guidelines is O the EEOC (Equal Employment Opportunity Commission). the OSHA (Occupational Safety and Health...
-
George owed Keith $800 on a personal loan. Neither the amount of the debt nor Georges liability to pay the $800 was disputed. Keith had also rendered services as a carpenter to George without any...
-
Which of the following accounting issues is most likely to raise an auditor's professional skepticism about earnings manipulation? a. Progress payments. b. Allowance for doubtful accounts. c. Sales...
-
The composite aluminum 2014-T6 bar is made from two segments having diameters of \(7.5 \mathrm{~mm}\) and \(15 \mathrm{~mm}\). Determine the maximum height \(h\) from which the 10-kg collar should be...
-
CLOSING INCOME SUMMARY AND DIVIDENDS TO RETAINED EARNINGS Davidson Company had a net income of $120,000 and paid cash dividends of $24,000 for 20--. Wilson and Picket Company had a net loss of...
-
(10%) Problem 10: Consider the point charges arranged at the corners and at the center of a square, as depicted in the figure. ab O O qa 090 Og Find the magnitude of the net Coulomb force, in...
-
Use data from Table 10.3 to estimate the enthalpy change ( r H) for the following reaction. Table 10.3 CH6(g) + Cl2(g) CH5Cl(g) + HCl(g) AH = ?
-
Write a Lewis structure of the hydroxylamine molecule, H 2 NOH. Then, with data from Table 10.2, determine all the bond lengths. Table 10.2 TABLE 10.2 Some Average Bond Lengthsa Bond Length, pm 74.14...
-
Sams Garage is trying to determine the cost of providing an oil change. Why would the average cost of this service be more relevant information than the actual cost for each customer?
-
You can cope with the pressures of your job in a healthy way if you have good stress management skills. After all it's not the thoughtless, angry, pushy customers or the endless complaints, questions...
-
The spot price of an asset is $500 and the risk-free rate for all maturities is 3% with continuous compounding. The asset provides an income of $20 in six months, and again in one year. What is...
-
Please watch this YouTube video on managing employee benefits https://www.youtube.com/watch?v=Ja9swkXpgGc Links to an external site. and respond to the following questions: Which elements of benefits...
-
During 2022, Oriole Company purchased 17700 shares of Whispering Winds Corp. common stock for $398250 as a passive interest investment. The fair value of these shares was $388515 at December 31,...
-
Solve lim (9x2+x-3x) 818
-
When projects have longer lives, it is more difficult to accurately estimate the cash flows and discount rates over the life of the project. Explain why this statement is true.
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
Respond to the following comments. a. My companys cost of capital is the rate we pay to the bank when we borrow money. b. Net present value is just theory. It has no practical relevance. We maximize...
-
Ms. Smith is retired and depends on her investments for retirement income. Mr. Jones is a young executive who wants to save for the future. They are both stockholders in Airbus, which is investing...
-
Answer this question by drawing graphs like Figure. Casper Milk toast has $200,000 available to support consumption in periods 0 (now) and 1 (next year). He wants to consume exactly the same amount...
-
a) What is IPv4? Show and explain the meaning of IPv4 elements: b) If you are given the IPv4 address 1001 1000 11011001 11000000 1100 1111, what is that address in dotted decimal notation? Show and...
-
Consider a computation running on a machine with a 1 GHz clock, 8-word cache line, single cycle access to the cache, and 100 ns latency to DRAM. The computation has a cache hit ratio at 1 KB of 40%...
-
A group of Computer Security students just started a new semester and are taking the Group Project subject. For the subject, students will build a small secure network which consists of four server...

Study smarter with the SolutionInn App


