Use data from Table 13.8 and Figure 13-10 to estimate the temperature at which K = 1.0
Question:
Use data from Table 13.8 and Figure 13-10 to estimate the temperature at which K = 1.0 x 106 for the reaction
![]()
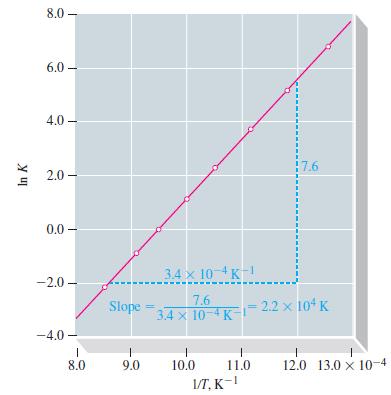
Table 13.8
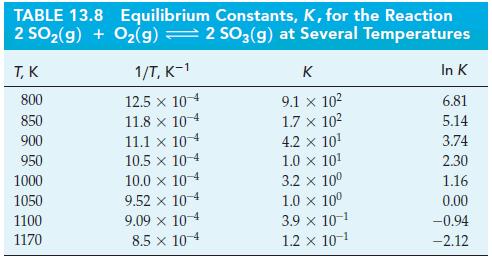
Figure 13-10
Transcribed Image Text:
2 SO2(g) + O2(g) - 2 SO3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Analyze By consulting Table 138 we see that K 91 x 10 ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) Estimate the temperature at which K = 5.8 x 10 -2 for the reaction in Example 13-12. Use data from Table 13.8 and Figure 13-10. (B) What is the value of K p for the reaction 2 SO 2 (g) + O 2 (g) ...
-
In Example 13-12, we used the vant Hoff equation to determine the temperature at which for the reaction 2 SO 2 (g) + O 2 (g) 2 SO 3 (g). Obtain another estimate of this temperature with data from...
-
Use data from Appendix D and other information from this chapter to estimate the temperature at which the dissociation of I 2 (g) becomes appreciable [for example, with the I 2 (g) 50% dissociated...
-
Use Eq. (6) to estimate Af = f(3.53, 8.98) - f(3.5,9)
-
Outsourcing by both manufacturers and service companies is becoming increasingly common. There are now many firms that specialize in outsourcing consulting. Address: www.alsbridge.com, or go to...
-
The molar mass of an enzyme was determined by dissolving it in water, measuring the osmotic pressure at 20C, and extrapolating the data to zero concentration. The following data were obtained: c/(mg...
-
How does an auditor test the valuation of accounts receivable and sales?
-
Several years after reengineering its production process, Trudell Corp. hired a new controller, Georgia Taylor. She developed an ABC system very similar to the one used by Trudells chief rival. Part...
-
The probability distribution for chlorine gas is shown in the diagram below. Determine the temperature of the gas. Take the molar mass of chlorine to be 80 g/mol. K P 580 1160 1740 2320 2900 v (m/s)
-
(A) Dinitrogen pentoxide, N 2 O 5 , is a solid with a high vapor pressure. Its vapor pressure at 7.5 C is 100 mmHg, and the solid sublimes at a pressure of 1.00 atm at 32.4 C. What is r G 25 C for...
-
By analogy to f H and f G how would you define standard entropy of formation? Which would have the largest standard entropy of formation: CH 4 (g), CH 3 CH 2 OH(l), or CS 2 (l)? First make a...
-
Why is Ricardo's explanation of the law of comparative advantage unacceptable? What acceptable theory can be used to explain the law?
-
Simplify (18xyz)/(6xy4)
-
Explain the dyamics that you would expect to find in a meadow/forest food web with grasses, deer and wolves if a fire killed all of the grass species one season? What steps could you take as a...
-
Punnett squares are a useful tool in genetic studies. Describes the most likely use of this tool?
-
What is the role of a regulatory gene?
-
1. Mohamed Salah formed a company named Salah Air Farming Ltd. In this company, he owned majority of the shares except one share which was held by his own wife; Mrs Salah. He was the company's sole...
-
Calculate the unit product cost using absorption costing. Round your answer to the nearest cent. Marks Company had the following costs: Units produced.................... 300 units Direct materials...
-
(a) Water flows through the nozzle of a garden hose. Find an expression for m in terms of line pressure P 1 , ambient pressure P 2 , inside hose diameter D 1 , and nozzle outlet diameter D 2 . Assume...
-
Kingsport Containers, Ltd, of the Bahamas experiences wide variation in demand for the 200-liter steel drums it fabricates. The leakproof, rustproof steel drums have a variety of uses from storing...
-
Vista Landscaping uses a job-order costing system to track the costs of its landscaping projects. The company provides garden design and installation services for its clients. The table below...
-
White Company has two departments, Cutting and Finishing. The company uses a job-order costing system and computes a predetermined overhead rate in each department. The Cutting Department bases its...
-
You are a mid-level management consultant working for the top tier global consulting firm McKinsey & Co. You have been engaged by a private equity client that has acquired a firm in a sector they...
-
According to a study published in February 2023 by the Chemical Engineering and Bioprocesses academic at the Catholic University (UC), Hctor Jorquera, who analyzed the air quality in Quintero and La...
-
Matilda Jacobs arrived at her Sydney offices at 6.30am. She glanced at her sales team's figures for the last quarter. There had been a noticeable decline in their performance. This puzzled her. In...

Study smarter with the SolutionInn App


