Use data from Table 7.2 to determine the standard heat of combustion of C 2 H 5
Question:
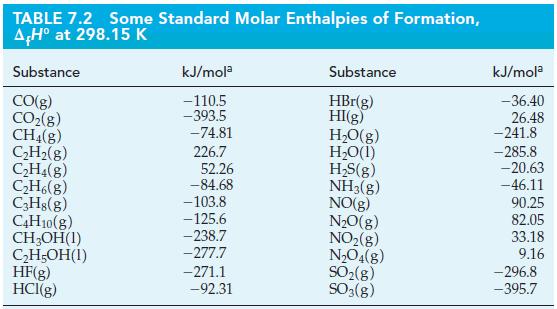
Use data from Table 7.2 to determine the standard heat of combustion of C2H5OH(l), if reactants and products are maintained at 25 °C and 1 bar.
Table 7.2
Transcribed Image Text:
TABLE 7.2 AH° at 298.15 K Substance CO(g) CO₂(g) CH₂(g) C₂H₂(g) C₂H4(g) C₂H6(g) C3H8(g) C4H10(g) CH3OH(1) C₂H5OH(1) Some Standard Molar Enthalpies of Formation, HF(g) HCI(g) kJ/mola -110.5 -393.5 -74.81 226.7 52.26 -84.68 -103.8 -125.6 -238.7 -277.7 -271.1 -92.31 Substance HBr(g) HI(g) H₂O(g) H₂O(1) H₂S(g) NH3(g) NO(g) N₂O(g) NO₂(g) N₂O4(g) SO₂(g) SO3(g) kJ/mola -36.40 26.48 -241.8 -285.8 -20.63 -46.11 90.25 82.05 33.18 9.16 -296.8 -395.7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Balanced chemical equation for the combustion ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The standard heat of combustion of liquid n-octane to form CO 2 and liquid water at 25C and 1 atm is H c = 5471 kJ/mol. (a) Briefly explain what that means. Your explanation may take the form When...
-
The standard heat of the combustion reaction of liquid n-hexane to form CO 2 (g) and H 2 O(l), with all reactants and products at 77F and 1 atm, is H r = 1:791 10 6 Btu. The heat of vaporization of...
-
In Problem 3.67 on page 135, you were introduced to a tea-bag-filling operation. An important quality characteristic of interest for this process is the weight of the tea in the individual bags. The...
-
Listed here are the total costs associated with the production of 10,000 Blu-ray Discs (BDs) manufactured by New Age. The BDs sell for $15 each. Required 1. Classify each cost and its amount as (a)...
-
Select Ltd is registered with an authorized capital of 300,000 ordinary shares of 1. The following trial balance was extracted from the books of the company on 31 March 2016, after the preparation of...
-
Diageo North America, Inc., the owner of the Bulleit brand family of whiskeys, sued competitor W.J. Deutsch & Sons Ltd. for allegedly infringing upon the trade dress of Diageos Bulleit bottles....
-
The controller of Sonoma Housewares Inc. instructs you to prepare a monthly cash budget for the next three months. You are presented with the following budget information: The company expects to sell...
-
. Imagine you have an on-premises eCommerce site for a small local jewelry store. . The jewelry store sells rings, watches, necklaces and bracelets. . Review Amazon Web Services, Microsoft Azure and...
-
In this mini-case you will perform some procedures required as a part of audit planning. For ease your audit manager has already organized the workpapers and completed several of the required...
-
Use data from Table 7.2, together with the fact that r H = -3509 kJ mol -1 for the complete combustion of pentane, C 5 H 12 (l), to calculate r H for the reaction below. Table 7.2 5 CO(g) + 11 H(g)...
-
Use data from Appendix D to calculate r H for the following reaction at 25 C. Fe2O3(s) + 3 CO(g) 2 Fe(s) + 3 CO(g) A,H = ?
-
Evaluate the given trigonometric functions directly, without first changing to degree measure. Round answers to three significant digits. sin 4.24
-
14 4 points A company reports the following amounts at the end of the year: Sales revenue Cost of goods sold Net income $360,000 240,000 53,000 Saved Compute the company's gross profit ratio. (Round...
-
John and Betty are married John's uncle Tom has lived with John and Betty since February 2022. Tom has gross income of only $4,000 for 2023 which he uses for his own support. John and Betty help with...
-
Tom has itemized deductions of $8,000 for 2023. He files single. His Adjusted Gross Income is $50,000. What is his tax ?
-
The ergodic Markov chain (Figure 1) is in steady state at time 0 and its states are given = {a, b, c). Write the equations for as S (1,2,3,4). The corresponding source alphabet is the steady-state...
-
find the present value of the given annuity. $ 533 per month for 36 months at the rate of 3.9 % compounded monthly
-
The inventory of Wood4Fun and data on purchases and sales for a two-month period follow. The company closes its books at the end of each month. It uses the periodic inventory system. Required 1....
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
Nonmonetary Exchanges Holyfield Corporation wishes to exchange a machine used in its operations. Holyfield has received the following offers from other companies in the industry. 1. Dorsett Company...
-
Nonmonetary Exchanges on August 1, Hyde, Inc. exchanged productive assets with Wiggins, Inc. Hydes asset are referred to below as Asset A, and Wiggins is referred to as Asset B. The following facts...
-
Nonmonetary Exchanges During the current year, Marshall Construction trades an old crane that has a book value of $90,000 (original cost $140,000 less accumulated depreciation $50,000) for a new...
-
The fish population in a certain lake rises and falls according to the formula 1000 (30+ 17t-) Here F is the number of fish at time t, where t is measured in years since January 12002, when the fish...
-
2. Bob bought some land costing $16,040. Today, that same land is valued at $46,117. How long has Bob owned this land If the price of land has been Increasing at 4 percent per year?
-
How can advances in genomics, single-cell sequencing, and live-cell imaging technologies be leveraged to elucidate the spatiotemporal dynamics of meiotic events, meiotic gene expression networks, and...
Introduction To The Analysis Of Electromechanical Systems 1st Edition - ISBN: 1119829992 - Free Book

Study smarter with the SolutionInn App


