Use data from the text to construct a standard electrode potential diagram relating the following vanadium species
Question:
Use data from the text to construct a standard electrode potential diagram relating the following vanadium species in acidic solution.
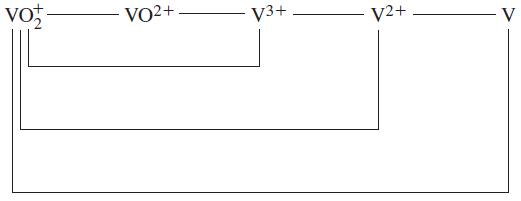
Transcribed Image Text:
ta vo VO2+ V3+ V2+ V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Based on the information provided in the image the following standard electrode potential diagram ca...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write a Lewis structure for each of the following organic molecules All the Diagrams are included in the Solution
-
Derive the form of the Longmuir isotherm for the complete dissociative adsorption of SO2, i.e.: Diagrams included in the Solution
-
Use data from the text to construct a standard electrode potential diagram relating the following chromium species in acidic solution. Cr0- Cr+ Cr2+ - Cr
-
Mary Sue owns 600 shares of QRS Moving Company. QRS pays a quarterly dividend of $0.50 per share. What is the total annual dividend that Mary Sue will receive?
-
On January 1, 2012, Hanks Company leased a copy machine with an integrated laser printer from Officeneeds, Inc. The five-year lease is noncancelable and requires monthly payments of $200 at the end...
-
In your own words, define the terms sustainability, sustainable management, and sustainability management accounting.
-
The Bonferroni adjustment is made by multiplying the P-value by the number of___________________ . In Exercises 3 and 4, fill in each blank with the appropriate word or phrase.
-
Callahan Manufacturing has assembled the data appearing below pertaining to two products. Past experience has shown that the unavoidable fixed factory overhead included in the cost per machine hour...
-
An annuity consists of 40 payments of $500 each made at intervals of 3 months. Interest is atj1= 4.5%. Determine the value of this annuity at each of the following times: (a) 3 months before the time...
-
When a soluble lead compound is added to a solution containing primarily orange dichromate ion, yellow lead chromate precipitates. Describe the equilibria involved.
-
The electrode potential diagram for manganese in acidic solutions in Figure 23-14 does not include a value of E for the reduction of MnO 4 to Mn 2+ . Use other data in the figure to establish this...
-
Given MA lever = SE/SR, find each missingquantity. MA 49.2 cm 29.7 in. 29.7 cm cm SE 76.3 cm 67.4 cm 12. 7.00 13 14. 4.00 67.4 cm
-
Doosan Babcock, a Scottish electronic equipment manufacturer and a publicly held company, is evaluating the cost of equity capital for the firm. The firms shares are selling for \($35.00;\) it...
-
You purchased a bond issued by Motor Oil Hellas for \($1,334.\) The bond has a coupon rate of 8.3 percent, which is paid semiannually. It matures in 16 years and has a par value of \($1,000.\) What...
-
The common stock for Beta Corporation sells for \($50.\) If a new issue is sold, the flotation costs are estimated to be 5 percent. The company pays 50 percent of its earnings in dividends, and a...
-
Refer to Googles financial statements in Appendix A to compute its equity ratio as of December 31, 2015, and December 31, 2014. Data From Google Financial Statement Appendix A Google Inc....
-
Assume the market price of a 7-year bond for Alma Telecom Services is \($985,\) and it has a par value of \($1,000\). The bond has an annual interest rate of 7 percent that is paid semiannually. What...
-
Dalton Brothers pay 15% in taxes on income between $1 and $40,000. All income above $40,000 is taxed at 40%. The firms variable costs as a percent of revenues are 60%. Annual fixed costs are...
-
The activities listed in lines 2125 serve primarily as examples of A) Underappreciated dangers B) Intolerable risks C) Medical priorities D) Policy failures
-
The following selected accounts appear in the adjusted trial balance for Cohen Company. Indicate the financial statement on which each account would be reported. (a) Accumulated Depreciation. (b)...
-
Using the data in BE4-12, identify the accounts that would be included in a post-closing trial balance.
-
The income statement for the Timberline Golf Club Inc. for the month ended July 31 shows Service Revenue $16,000; Salaries and Wages Expense $8,400; Maintenance and Repairs Expense $2,500; and Income...
-
Assume that Stillwater Designs produces two automotive subwoofers: S12L7 and S12L5. The S12L7 sells for $475, and the S12L5 sells for $300. Projected sales (number of speakers) for the coming five...
-
How does organizational learning contribute to continuous improvement and adaptability in a rapidly changing business environment ?
-
You are required to pick a current ("hot", widely discussed) economic event that relates to the material (topic) we have covered or will cover in this course. This paper has to show your ability to...

Study smarter with the SolutionInn App


