Use Figure 23-8 to estimate for the reaction ZnO(s) + C(s) Zn(l) + CO(g), at 800
Question:
Use Figure 23-8 to estimate for the reaction ZnO(s) + C(s) ⇌ Zn(l) + CO(g), at 800 °C,
(a) A value of Kp;
(b) The equilibrium pressure of CO(g).
Figure 23-8
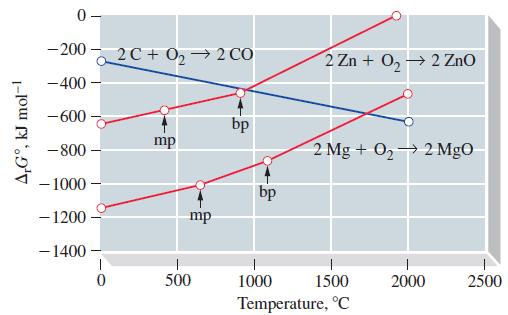
Transcribed Image Text:
A,Gº, kJ mol-1 0- -2002C+0₂ → 2 CO - 400 -600 -800- - 1000 - 1200 - 1400 0 mp mp 500 bp bp 2 Zn + 0₂ → 2 ZnO 2 Mg + O₂ 2 MgO 1000 1500 Temperature, °C 2000 2500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a To estimate a value of Kp for the reaction ZnOs Cs Znl COg at 800 C using Figure 238 we need to fi...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Use average bond enthalpies (Table 8.4) to estimate for the atomization of benzene, C6H6: C6H6 (g) 6C (g) + 6 H (g) Compare the value to that obtained by using Hof data given in Appendix C and...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-7. Ivan sold the following securities during the year and received a Form 1099-B that...
-
I am learning intermediate accounting and how to account for leases. I have a question about the calculation of present value for the right to use of the lease. my example specifically has a...
-
Is net income under- or overstated if inventory is sold and shipped but not recorded as a sale?
-
What is the difference between an open-end mutual fund and a closed-end fund? What is the difference between an open-end mutual fund and a unit investment trust?
-
How can you use a basic search on the Internet to help you in your informal fact gathering and investigation?
-
Review the Comprehensive Annual Financial Report (CAFR) that you obtained. 1. What are three main sections of the report? 2. Review the introductory section of the CAFR. a. Was the entitys annual...
-
Determine the amount of cost-sharing that must be paid by the patient in the scenario below. Ava received a service on January 1st, and the bill was $900. She has a deductible of $1,000 and a...
-
At 400 C, r G = 25 kJ mol 1 for the reaction 2 Hg(l) + O 2 (g) 2 HgO(s). If a sample of HgO(s) is heated to 400 C, what will be the equilibrium partial pressure of O 2 (g)?
-
Use the result of the Integrative Example to determine whether a solution can be prepared with [Cu + ] equal to (a) 0.20 M; (b) 1.0 x 10 10 M. Integrative Example Although a number of slightly...
-
J. Barnes, CPA, has been retained to audit a manufacturing company with a balance sheet that includes the caption Property, Plant, and Equipment. Barnes has been asked by the company's management if...
-
The period of a pendulum on the surface of the Earth is 1. It is then brought at a higher altitude. For it to maintain its period, should the length of the pendulum string be made longer or shorter?
-
A mass attached to a spring oscillates in simple harmonic motion along the \(x\)-axis. The limits of its motion are \(x=-5 \mathrm{~cm}\) and \(x=45 \mathrm{~cm}\) and it goes from one of these...
-
A certain metallic structure weighs \(2450 \mathrm{~N}\) on earth. The structure is sent to Mars, which has a diameter of \(6.78 \times 10^{6} \mathrm{~m}\) and mass of \(6.42 \times 10^{23}...
-
Two spherical celestial bodies, one of mass \(3.5 \times 10^{13} \mathrm{~kg}\) and the other of mass \(2.4 \times 10^{18} \mathrm{~kg}\), experience a gravitational force between them with a...
-
A battery and four identical light bulbs are arranged in the circuit of Figure P31.3. Rank the current magnitudes at the nine lettered locations from greatest to smallest. Data from Figure P31.3 90 e...
-
This semester, you moved to an apartment eight miles from campus and will commute to classes three times a week. This decision will let you achieve significant savings. However, you have not yet...
-
A parking lot charges $3 for the first hour (or part of an hour) and $2 for each succeeding hour (or part), up to a daily maximum of $10. (a) Sketch a graph of the cost of parking at this lot as a...
-
The financial statements of Zetar plc are presented in Appendix C. The companys complete annual report, including the notes to its financial statements, is available at www.zetarplc.com. Instructions...
-
Emily Frazier believes a current liability is a debt that can be expected to be paid in one year. Is Emily correct? Explain.
-
Verona Company obtains $20,000 in cash by signing a 9%, 6-month, $20,000 note payable to First Bank on July 1. Veronas fiscal year ends on September 30. What information should be reported for the...
-
S dx Evaluate ( +1) x(x+1)
-
Morganton Company makes one product and it provided the following information to help prepare the master budget: a. The budgeted selling price per unit is $65. Budgeted unit sales for June, July,...
-
1.Taxpayer, a cash method, calendar year taxpayer, engaged in the following transactions in shares of stock. Consider the amount and character of T's gain or loss in each transaction: (a)T bought 100...

Study smarter with the SolutionInn App


