What is the pressure (in mmHg) of the gas inside the apparatus below if P bar. =
Question:
What is the pressure (in mmHg) of the gas inside the apparatus below if Pbar. = 740 mmHg, h1 = 30 mm, and h2 = 40 mm?
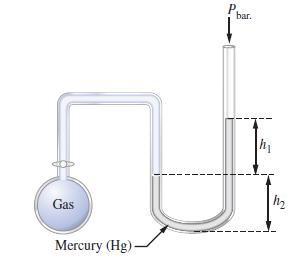
Transcribed Image Text:
Gas P bar. h₁ H Mercury (Hg) m₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To determine the pressure of the gas inside the apparatus we need to consid...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
In Figure P2.4.7, a single-reading mercury manometer is used to measure water pressure in the pipe. What is the pressure (in psi) if h1 = 6.9 in and h2 = 24.0 in.? h2 Figure P2.4.7
-
What is the pressure in kPa inside a container if it holds water that is just beginning to boil at a) 140C, b) 200C, c) 320C?
-
The atmospheric pressure at the summit of Mt. McKinley is 606 mmHg on a certain day. What is the pressure in atm and in kPa?
-
In Exercise solve the given equations and check the results. F-3 12 2 3 || 1 - 3F 2
-
Define expectancy theory.
-
Wadkins Company, a machinery dealer, leased a machine to Aoki Corporation on January 1, 2015. The lease is for an 8-year period and requires equal annual payments of 38,514,000 at the beginning of...
-
Bert C. Roberts Jr. was chairman of WorldComs board of directors. Immediately before that, he had been chairman of MCI, which WorldCom acquired on September 14, 1998, in a transaction valued at...
-
The adjusted trial balance of Elmo Real Estate Appraisal at June 30, 2016, follows: Requirements 1. Prepare the companys income statement for the year ended June 30, 2016. 2. Prepare the companys...
-
Criticism of the World Bank is generally on a diverse range of issues but they generally centre around concern about the approaches adopted by the World Bank in formulating their policies, and the...
-
(A) A 1.00 mL sample of N 2 (g) at 36.2 C and 2.14 atm is heated to 37.8 C, and the pressure changed to 1.02 atm. What volume does the gas occupy at this final temperature and pressure? (B) Suppose...
-
What is the pressure, in kilopascals, exerted by 1.00 x 10 20 molecules of N 2 in a 305 mL flask at 175 C?
-
You want to start a company that makes dolls with ethnic features for Hispanic, Asian, and multiracial markets. Discuss how you would analyze your competitors benefits and develop a plan to gather...
-
One of the most successful collection programs is the 50 State Quarters program developed by the U.S. Mint. This program began in 1999 and permitted each state to put a design on the back of the U.S....
-
Explain with detail the basic principle underlying the law of tort? Individuals should always provide cases to keep the courts busythe opportunity to seek compensation for harm they have suffered a...
-
Explain capital gains tax, and also discuss whether the receipt of the designer watch constitutes assessable income.? Where appropriate, support your answer with legislative and case authority....
-
20 N at 30cm,30 N at 60 cm, and 10 N at 100 cm from the fixed end are the cantilever beam. The deflection under 30N load due to all loads is 2 mm, what would be the natural frequency of transverse...
-
Sharp Company manufactures a single product. Annual production costs incurred in the manufacturing process are shown below for the production of 2,000 units. Utilities is a mixed cost. The fixed...
-
A consumers budget set for two goods (X and Y) is 600 3X + 6Y. a. Illustrate the budget set in a diagram. b. Does the budget set change if the prices of both goods double and the consumers income...
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
(Basic Accounting for Temporary Differences) Dexter Company appropriately uses the asset liability method to record deferred income taxes. Dexter reports depreciation expense for certain machinery...
-
(Identify Temporary Differences and Classification Criteria) The asset-liability approach for recording deferred income taxes is an integral part of generally accepted accounting principles. (a)...
-
(Accounting and Classification of Deferred Income Taxes) Part A This year Gumowski Company has each of the following items in its income statement. 1. Gross profits on installment sales. 2. Revenues...
-
8. MgO has the same ionic crystal structure as NaCl shown below. The atomic radius for the Mg ion is .65A and the O ion is 1.4A. The Mg mass is 24.3g/mol and O is 16g/mol. (a) What is the packing...
-
1) Why do we have methane release from frequent flooding of the Amazon basin? because the water 2) Methane is a much more potent green house gas than CO2 - why?
-
1. A sample of polyisobutylene (PIB) has a glass transition temperature of 5C. At what frequency o would the energy lost per cycle in this PIB at 0C be same as it is at 1 Hz at 30C? (Note: You may...

Study smarter with the SolutionInn App


