What type of orbital (i.e., 3s, 4p, . . . ) is designated by these quantum numbers?
Question:
What type of orbital (i.e., 3s, 4p, . . . ) is designated by these quantum numbers?
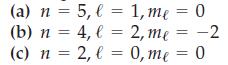
Transcribed Image Text:
(a) n = 5, l = 1, me = 0 (b) n = 4, l = 2, me = -2 (c) n = 2, l = 0, me = 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The quantum numbers provided can be used to determine the type of orbital The quantum numbers are in ...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
If one were to try to draw the simplest Lewis structure for molecular oxygen, the result might be the following However, it is known from the properties of molecular oxygen and experiments that O2...
-
Answer the following questions as a summary quiz on this chapter. (a) The quantum number n describes the __________ of an atomic orbital, and the quantum number describes its __________. (b) When n...
-
Do the three 4p atomic orbitals possess the same or different values of (a) Principal quantum number, (b) The orbital quantum number and (c) The magnetic quantum number? Write down a set of quantum...
-
Hi, the task is to critically evaluated two or more types of market segmentation, and applied to own organisations customer base. The guidance says to start with a general explanation of the topic -...
-
What could AIG have done differently to prevent its failure and subsequent bailout? When American International Group (AIG) collapsed in September 2008 and was subsequently saved by a government...
-
An inventor claims to have developed a refrigeration system that removes heat from the closed region at -12C and transfers it to the surrounding air at 25C while maintaining a COP of 6.5. Is this...
-
Jupiter's is considering an investment in time and administrative expense on an effort that promises one large payoff in the future, followed by additional expenses over a 10-year horizon. The cash...
-
River Companys chart of accounts includes the following selected accounts. 101 Cash 401 Sales Revenue 112 Accounts Receivable 414 Sales Discounts 120 Inventory 505 Cost of Goods Sold 301 Owners...
-
Write down a regular expression for the following language: L= {w (p, q): number of p's in w is divisible by 4 }
-
Randy Corp. issued $200,000 of 7.6% (payable each 28 February and 31 August), 4-year bonds. The bonds were dated 1 March 20X4, and mature on 28 February 20X8. The bonds were issued (to yield 8%) on...
-
Concerning the electrons in the shells, subshells, and orbitals of an atom, how many can have mg 1, and m = +- ? (a) n = 4, = 2, me = 1, and (b) n = 4, l = 2, and me = 1? (c) n = 4 and l = 2? (d) n =...
-
Write an acceptable value for each of the missing quantum numbers. (a) n = 3, (b) n (c) n = 4, l = (d) n = ?, l= ?, l = ?, me = 2, ms 2, me = 1, ms 2, me 0, me = = 0, ms ?, ms ? = ? 1 1 2
-
Describe the role of meetings at Sound in Motion.
-
Prepare a worksheet of a data given below. c. Prepare a three-part worksheet as of December 31, 20X5. Note: Values in the first two columns (the "parent" and "subsidiary" balances) that are to be...
-
2. What does the following divide and conquer Python program do? def solve(a, low, high): if low == high: return 0 else: mid = (low + high) // 2 #floor division x = min(a[low:mid+1]) #min element in...
-
Translate this loop into assembly code. int A = 3; int B = 10; while (true) { A++; B--; if (A
-
Analyze DISH Network DISH Network Corporation (DISH) provides satellite-based entertainment services to residential and business customers. Services are billed and collected on a monthly basis. DISH...
-
4. What will be the output of the following program: 1 int i = 43, j = -17; 2 do { 3 printf("%d", i+j); 4 j--; 5 } 6 while (0)
-
The units of an item available for sale during the year were as follows: There are 23 units of the item in the physical inventory at December 31. The periodic inventory system is used. Determine the...
-
The diagram shows the two forces acting on a small object. Which of the following is the resultant force on the object? A. 8 N downwards B. 8 N upwards C. 2 N downwards D. 2 N upwards 3 N 5 N
-
Based on what you can tell from the case, make a quick chart of Anglos organization prior to Carrolls changes. Then make a similar chart of its organization after those changes. Be prepared to...
-
Describe the results of Anglos changes to its organizational structure in terms of configuration, operational aspects, and responsibility and authority.
-
In addition to seven business units, Anglos restructuring creates five functional groupsFinance, Mining & Technology, Business Performance & Projects, HR & Communications, and Strategy & Business...
-
I have 720 grams of chemical A and mix it with 80 grams of chemical B. I accidentally put 730 grams of chemical A in. How much more of chemical B do I need to add to keep the same ratio?
-
POST: Using your results from 'Assessing Your Styles' on p. 153, compare your scores on the styles measure for Situation A & Situation B. Title your post either "same style both situations" or...
-
Follow the steps in the document, then attach the resultant file. Install a VMWare instance of Kali Linux or use a pre-built VM, link listed below. Add a username of 1, password of user402! Log in as...

Study smarter with the SolutionInn App


