Which of the following represents a nonvolatile solute? Gas Aqueous Gas Aqueous Gas Aqueous (a) (b) Gas
Question:
Which of the following represents a nonvolatile solute?
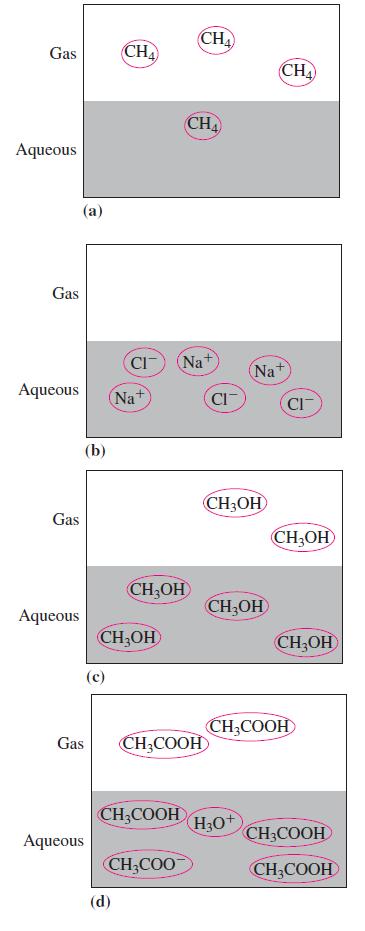
Transcribed Image Text:
Gas Aqueous Gas Aqueous Gas Aqueous (a) (b) Gas Aqueous CHA Na+ (d) CIT Na CH₂OH CH₂OH CH₂COOH CH₂ CH₂COO CHA CH₂COOH CI CH3OH Na+ CH3OH CHA H3O+ CI CH₂OH CH₂OH CH₂COOH CH3COOH CH3COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The only nonvolatile solute is c CH3OH The other options are all volatile solutesA volatile solu...View the full answer

Answered By

Ajeet Singh
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life.
I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge.
I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields.
Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a teacher. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
4.90+
7+ Reviews
15+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
According to Table 1, the concentrations of which of the following substances varies the least with temperature? A solute is any substance that is dissolved in another substance, which is called the...
-
According to Table 1, HCl would most likely have which of the following concentrations at 70 C? A solute is any substance that is dissolved in another substance, which is called the solvent. A...
-
Which of the following represents a firms average gross receivables balance? I. Days sales in receivables x accounts receivable turnover. II. Average daily sales x average collection period. III. Net...
-
Apply the product rule for exponents, if possible. y 4 y 5 y 6
-
During the year, prepaid expenses decreased $6,500, and accrued expenses increased $2,000. Indicate how the changes in prepaid expenses and accrued expenses payable should be entered in the...
-
A 1-megabit computer memory chip contains many 60.0-f F capacitors. Each capacitor has a plate area of 21.0 x 10-12 m2. Determine the plate separation of such a capacitor (assume a parallel-plate...
-
How do some online businesses determine the prices they will charge to individual customers?
-
Southwest Sand and Gravel paid $800,000 to acquire 1,000,000 cubic yards of sand reserves. The following statements model reflects Southwest's financial condition just prior to purchasing the sand...
-
Define Continuous Data Protection (CDP) and discuss how it differs from traditional backup approaches. What are the advantages and disadvantages of implementing CDP ?
-
In a saturated solution at 25 C and 1 bar, for the following solutes, which condition will increase solubility? (a) Ar(g), decrease temperature; (b) NaCl(s), increase pressure; (c) N 2 , decrease...
-
When NH 4 Cl dissolves in a test tube of water, the test tube becomes colder. Is the magnitude of H lattice for NH 4 Cl larger or smaller than the sum of H hydration of the ions?
-
Prove Eq. (9.4.14) in Example 9.4.6. Both parts of the integrand in Eq. (9.4.13) differ from gamma distribution p.d.f.s by some factor that does not depend on t.
-
G & L Plastic Molders paid consulting fee of $73,000 last week for feedback on a new project. This week the company is trying to decide whether the new project should be undertaken. Should the...
-
Amazon (AMZN) began trading in mid-1997. At the end of 1997, AMZN's stock price was around $4. At the end of 2021, AMZN was trading at $3334. What is the annualized rate of return on AMZN stock?
-
Asia-based Ashraf Inc., a high-tech company, raised $92 million in its initial public offering (IPO). Of the $35 offering price per share, the corporation got $29. $400,000 was spent on the firm's...
-
1. What management approach does Riverside State Hospital's style of man- agement most resemble? 2. How does the management approach influence communication at the hospital? 3. Should Ellis be held...
-
In the event of liquidation, HPI preferred stockholders are entitled to $ 3 0 . 5 0 plus accrued dividends. Does this mean that preferred stockholders will receive that amount? Discuss.
-
Identify the most important accounts, management assertions, and risks for each of the following industries:
-
Repeat the previous problem, but close the positions on September 20. Use the spreadsheet to find the profits for the possible stock prices on September 20. Generate a graph and use it to identify...
-
Define the market capitalization rate for a stock. Does it equal the opportunity cost of capital of investing in the stock?
-
Rework Table 4.1 under the assumption that the dividend on Fledgling Electronics is $10 next year and that it is expected to grow by 5 percent a year. The capitalization rate is 15percent. Expected...
-
In March 2001, Fly Papers stock sold for about $73. Security analysts were forecasting a long-term earnings growth rate of 8.5 percent. The company was paying dividends of $1.68 per share. a. Assume...
-
You will write a report (4-5 pages) on any ONE of the following topics: a. Summarize the corporate social responsibility of any company of your choice. b. Evaluate three programs offering training...
-
TETLEY TEA CANADACanadian Tea Challenge Executive Summary (Written Report Only) The executive summary should include a concise statement of the problem, a short summary of the major points arising...
-
Identify the 8 elements of communication process. Does feedback indicate that communication has been successful?

Study smarter with the SolutionInn App


