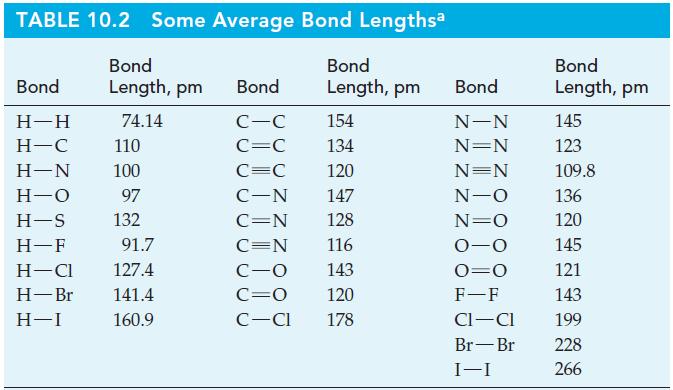
Write a Lewis structure of the hydroxylamine molecule, H 2 NOH. Then, with data from Table 10.2,
Question:
Write a Lewis structure of the hydroxylamine molecule, H2NOH. Then, with data from Table 10.2, determine all the bond lengths.
Table 10.2
Transcribed Image Text:
TABLE 10.2 Some Average Bond Lengthsa Bond Length, pm 74.14 Bond H-H H-C H-N H-O H-S H-F 110 100 97 132 91.7 H-Cl 127.4 H-Br 141.4 H-I 160.9 Bond C-C C=C C=C C-N C=N C=N Bond Length, pm 154 134 120 147 128 116 C-O 143 C=O 120 C-Cl 178 Bond N-N N=N N=N N-O N=O 0-0 O=O F-F CI-CI Br-Br I-I Bond Length, pm 145 123 109.8 136 120 145 121 143 199 228 266
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
H H Calculate the bond length pm for NH NO OH using table values Solut...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The structure of borazine, B 3 N 3 H 6 , is a six-membered ring of alternating B and N atoms. There is one H atom bonded to each B and to each N atom. The molecule is planar. (a) Write a Lewis...
-
Sulfur tetra-fluoride (SF4) reacts slowly with O2 to form sulfur tetra-fluoride monoxide (OSF4) according to the following unbalanced reaction: SF4 (g) + O2 (g) OSF4 (g) The O atom and the four F...
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
ABC Insurance Company has issued a commercial package policy to the Henderson Company. ABC recently discovered that company executives misrepresented important information about the business to...
-
(a) Judy orally promises her daughter, Liza, that she will give her a tract of land for her home. Liza, as intended by Judy, gives up her homestead and takes possession of the land. Liza lives there...
-
Squeaky Clean, a commercial laundry service, has two clients, Super 6 Motel and Seaside Inn. The following is information about Squeakys revenues and costs (in thousands) by customer for the previous...
-
A sample of size 15 is drawn from a normal population. The sample standard deviation is s1 = 5.2. A sample of size 10 is drawn from another normal population. The sample standard deviation is s2 =...
-
A statement of financial affairs created for an insolvent corporation that is beginning the process of liquidation discloses the following data (assets are shown at net realizable values): Assets...
-
Wilmington Company has two manufacturing departments-Assembly and Fabrication. It considers all of its manufacturing overhead costs to be fixed costs. The first set of data that is shown below is...
-
A reaction involved in the formation of ozone in the upper atmosphere is O 2 2 O. Without referring to Table 10.3, indicate whether this reaction is endothermic or exothermic. Explain. Table 10.3...
-
Refer to the Integrative Example. Use data from the chapter to estimate the length of the NF bond in FNO 2 . Integrative Example Nitryl fluoride is a reactive gas useful in rocket propellants. Its...
-
Geoff Payne painted four houses during April 2010. For these jobs, he spent $1,200 on paint, $80 on mineral spirits, and $300 on brushes. He also bought two pairs of coveralls for $100 each; he wears...
-
1. Perfectly Competitive Markets a. Four kinds of market structure and characteristics for each kind b. Characteristics of a perfectly competitive market c. Price Takers d. MR DARP e. Perfectly...
-
Expanding the discourse to encompass the transformative potential of central bank digital currencies (CBDCs) in reshaping the monetary landscape and redefining the role of traditional banking...
-
The boss wants the Current Rates sheet to indicate whether each cabin has a fireplace and/or a hot tub. Columns G and H have been created to contain that data, but nobody knew how to make text...
-
Your current income is $50,000 per year, and you would like, when you retire, to maintain your current standard of living (i.e., your purchasing power). If you expect to retire in 30 years and expect...
-
Find derivative of the following functions and value of y =(1-2t)y, y(0) = -1/6
-
Are some process structures inherently safer or more environmentally friendly than others?
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
We said that maximizing value makes sense only if we assume well-functioning capital markets. What does well-functioning mean? Can you think of circumstances in which maximizing value would not be in...
-
Why is a reputation for honesty and fair business practice important to the financial value of the corporation?
-
It is sometimes argued that the NPV criterion is appropriate for corporations but not for governments. First, governments must consider the time preferences of the community as a whole rather than...
-
Suppose that you borrow $17,000 for a new car. You can select one of the following loans, each requiring regular monthly payments: Installment Loan A: three-year loan at 5.7% Installment Loan B:...
-
1. Open a new Excel workbook. 2. Create a table with the following columns: "Item," "Quantity," "Price," and "Total Cost." 4 marks 3. In the "Item" column, list five different items that you might...
-
Garcia Company has two operating departments (Phone and Earbuds) and one service department (Office). Its departmental income statements follow. Indirect expenses and service department expenses...

Study smarter with the SolutionInn App


