A 3.75-g sample of iron ore is transformed to a solution of iron(II) sulfate, FeSO 4 ,
Question:
A 3.75-g sample of iron ore is transformed to a solution of iron(II) sulfate, FeSO4, and this solution is titrated with 0.150 M K2Cr2O7 (potassium dichromate). If it requires 43.7 mL of potassium dichromate solution to titrate the iron(II) sulfate solution, what is the percentage of iron in the ore? The reaction is
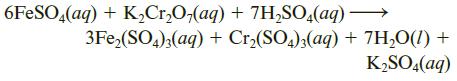
Transcribed Image Text:
6FESO,(aq) + K,Cr,O,(aq) + 7H,SO.(aq) → 3Fe,(SO,);(aq) + Cr,(SO,);(aq) + 7H,O(1) + K2SO,(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
First find the mass of Fe 2 required to react with the K 2 Cr 2 O 7 Pe...View the full answer

Answered By

Sabirah Shuaybi
I have diverse professional experience in teaching and tutoring - with all varieties of subjects ranging from Statistics, Computer Science/Programming, English as a Foreign Language and more. I am suited to working with all ages, from as young as 5 year olds to all the way up to the college level. I am looking forward to working with you and helping you achieve success in these subjects!
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A 3.33-g sample of iron ore is transformed to a solution of iron(II) sulfate, FeSO4, and this solution is titrated with 0.150 M K2Cr2O7 (potassium dichromate). If it requires 43.7 mL of potassium...
-
A sample of iron ore (containing only Fe2+ ions) weighing 0.2792 g was dissolved in dilute acid solution, and all the Fe(II) was converted to Fe(III) ions. The solution required 23.30mL of 0.0194 M...
-
A solution of hydrogen peroxide, H2O2, is titrated with a solution of potassium permanganate, KMnO4. The reaction is 5H2O2(aq) + 2KMnO4(aq) + 3H2SO4(aq) 5O2(g) + 2MnSO4(aq) + K2SO4(aq) + 8H2O(l ) It...
-
In Exercises find the indefinite integral. sech(2x1) dx
-
Several years ago, a growing number of warranty claims on Firestone tires sold on Ford SUVs prompted Firestone and Ford to issue a major recall. An analysis of warranty claims data helped identify...
-
The following is a list of terms and phrases discussed in the chapter. 1. Contra asset account 2. Permanent accounts 3. Depreciation 4. Adjusting entries 5. Prepaid expenses (prepayments) 6....
-
A card is randomly selected from a standard deck of 52 playing cards. Find the probability that the card is between 7 and 10, inclusive, or is black. Find the probability.
-
Why is organizational diagnosis essential to the success of any change effort?
-
The balance sheet for Fourth Corp. is shown here in market value terms. There are 5,000 shares of stock outstanding. Market Value Balance Sheet Cash $ 45,100 Equity 495,100 $ Fixed 450,000 assets...
-
Walkers is planning to acquire Cayman Bank, a freestanding C corporation, in expectation that new management can be brought in to achieve substantial operating efficiencies. You have been retained to...
-
How many milliliters of 0.250 M KMnO 4 are needed to react with 3.55 g of iron(II) sulfate, FeSO 4 ? The reaction is as follows: 10FESO,(aq) + 2KMNO,(aq) + 8H,SO,(aq) 5Fe,(SO4);(aq) + 2MNSO,(aq) +...
-
Nickel(II) sulfate solution reacts with sodium hydroxide solution to produce a precipitate of nickel(II) hydroxide and a solution of sodium sulfate. Write the molecular equation for this reaction....
-
Starting with the beginning balances in M2-9 and given the transactions in M2-5 (including the sample), prepare a balance sheet for Dennen, Inc., as of January 31, 2015, classified into current and...
-
What is the output of the following code segment? total = 0 for count in range(4,2,-1): total+count print(total)
-
XYZ, Inc. just paid dividend of $10.98. The dividends are expected to grow at 5.81% each year forever. The required rate of return on the stock is 14.04%. What is today's price of the stock?
-
Your investment portfolio consists entirely Google stock. Suppose that the risk-free rate is 4%, Google stock has an expected return of 14% and a volatility of 35%, and the market portfolio has an...
-
You are working as a marketing coordinator for a smart phone manufacturer. You have just realised that your manager is not familiar with the product life cycle stages. To address this issue, you need...
-
Camping Company manufactures camping tents from a lightweight synthetic fabric. Each tent uses the following standard material and labor costs. Direct Labor Direct Material Standard cost per tent 2...
-
How could managers from Japan or other Asian cultures adopt practices from the U.S. and European cultures when investing in those regions?
-
What are bounds and what do companies do with them?
-
A block of aluminum and a block of iron, both having the same mass, are removed from a freezer and placed outside on a warm day. When the same quantity of heat has flowed into each block, which block...
-
You have two samples of different metals, metal A and metal B, each having the same mass. You heat both metals to 95oC and then place each one into separate beakers containing the same quantity of...
-
Consider the reactions of silver metal, Ag(s), with each of the halogens: fluorine, F2(g), chlorine, Cl2(g), and bromine, Br2(l). What chapter data could you use to decide which reaction is most...
-
With regard to the production possibilities frontier (PPF), an efficient point is a point that is: ?
-
Within the context of rapidly evolving regulatory landscapes and heightened ethical scrutiny, how do advanced organizations navigate the ethical complexities of technological innovation and...
-
Write a program that accepts an integer limit and an integer value and that prints a complete line of output reporting the multiples of value uptil the limit . Keep prompting the user for a value...

Study smarter with the SolutionInn App


