How many milliliters of 0.250 M KMnO 4 are needed to react with 3.55 g of iron(II)
Question:
How many milliliters of 0.250 M KMnO4 are needed to react with 3.55 g of iron(II) sulfate, FeSO4? The reaction is as follows: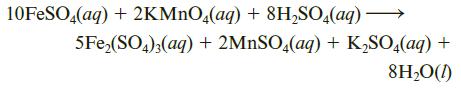
Transcribed Image Text:
10FESO,(aq) + 2KMNO,(aq) + 8H,SO,(aq) → 5Fe,(SO4);(aq) + 2MNSO,(aq) + K,SO,(aq) + 8H,O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The reaction is 10FeSO4 2KMnO4 8HSO...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many milliliters of 0.250 M KMnO4 are needed to react with 3.36 g of iron(II) sulfate, FeSO4? The reaction is as follows: 10FeSO4(aq) + 2KMnO4(aq) + 8H2SO4(aq) 5Fe2(SO4)3(aq) + 2MnSO4(aq) +...
-
For Reaction 1-7, how many milliliters of 0.165 0 M KMnO4 are needed to react with 108.0 mL of 0.1650 M oxalic acid? How many milliliters of 0.1650 M oxalic acid are required to react with 108.0 mL...
-
How many milliliters of 3.00 M H2SO4 are required to react with 4.35 g of solid containing 23.2 wt% Ba(NO3)2 if the reaction is Ba2++ SO24 BaSO4(s)?
-
In Exercises verify the identity. coshx = 1 + cosh 2x 2
-
One of the major measures of the quality of service provided by an organization is the speed with which the organization responds to customer complaints. A large family-held department store selling...
-
Greenock Limited has the following information available for accruals for the year ended December 31, 2022. The company adjusts its accounts annually. 1. The December utility bill for $425 was...
-
A 10-sided die, numbered 1 to 10, is rolled. Find the probability that the roll results in an even number or a number greater than 6. Use the pie chart at the left, which shows the percent...
-
Refer to Cornerstone Exercise 8.13. In March, Nashler Company produced 163,200 units and had the following actual costs: Direct materials..... $1,170,000 Direct labor ........ 258,000 Supplies...
-
Discuss the role of human factors in hazard analysis. How do techniques like Human Reliability Analysis (HRA) contribute to understanding and mitigating risks associated with human errors in...
-
CT Gift Shop is profitable but has a cash flow problem as of the end of June. December is the busiest month, and the shop expects to make a decent profit by the end of the year. As of the end of...
-
How many milliliters of 0.250 M H 2 SO 4 (sulfuric acid) are required to react with 8.20 g of sodium hydrogen carbonate, NaHCO 3 , according to the following equation? H 2 SO 4 (aq) + 2NaHCO 3 (aq) h...
-
A 3.75-g sample of iron ore is transformed to a solution of iron(II) sulfate, FeSO 4 , and this solution is titrated with 0.150 M K 2 Cr 2 O 7 (potassium dichromate). If it requires 43.7 mL of...
-
Conoco Phillips announced the purchase of 7.6% of the stock of Lukoil (a largely government-owned Russian oil and gas company) for $2.36 billion during a government auction of Lukoils stock. Conoco...
-
G1 G2 G3 G4 Demand C1 3 4 4 3 30 C2 4 3 2 1 45 C3 2 5 5 7 55 A. C4 333 6 1 9 3 25 OP C5 7 3 50 Capacity 45 60 75 35 Work out a transportation plan so as to minimize the transportation cost and find...
-
ABC. Inc just paid a dividend of $2.58 per share. The dividends are expected to increase by 1% each year. The required rate of return on the stock is 23%. What is the stock's expected price 9 years...
-
On July 1 , the supplies account balance was $ 1 , 6 6 5 . During July, supplies of $ 3 , 7 9 0 were purchased, and $ 1 , 7 0 4 of supplies were on hand as of July 3 1 . Determine supplies expense...
-
What is the difference between a stock's current market price and its intrinsic value? What is free cash flow? If you were an investor, why might you be more interested in free cash flow than net...
-
A stock is currently priced at $20. We model the possible movement of stock price over the next 3 months using a one-step Binomial tree. This means that u = 1.1618 and d = 0.8607. The riskfree rate...
-
The Downer family has owned Downer's Dairy Farm for generations. The farm totals approximately 800 acres of land. Until this year, John and his oldest son, Jack, managed the farm. John, a widower,...
-
When is the indirect pattern appropriate, and what are the benefits of using it?
-
A 250-g sample of water at 20.0oC is placed in a freezer that is held at a constant temperature of 20.0oC. Considering the water as the system, answer the following questions: a. What is the sign of...
-
A 20.0-g block of iron at 50.0oC and a 20.0 g block of aluminum at 45oC are placed in contact with each other. Assume that heat is only transferred between the two blocks. a. Draw an arrow indicating...
-
What is the enthalpy change for the preparation of one mole of liquid water from the elements, given the following equations? H2(g) +-02(g)--H20(g):AH, H20(1)--H20(g); ,up
-
Pharoah Enterprises purchased a delivery truck on January 1 , 2 0 2 5 , at a cost of $ 2 6 , 0 0 0 . The truck has a useful life of 7 years with an estimated salvage value of $ 5 , 9 1 0 . The...
-
Consider the following recurrence relation: A(1)=1, A(n) = 2A(n-1)+2"-1 Use the method of unraveling to find a closed form for A(n).
-
Given the following formula: Monthly Pay= [rate + rate / ([1+rate] ^(months) -1) X principle Where rate of 6% means 6/1200 and Months means number of years x 12 Possible data to test: Principle 12200...

Study smarter with the SolutionInn App


