One of the following compounds has a carbonnitrogen bond length of 116 pm; the other has a
Question:
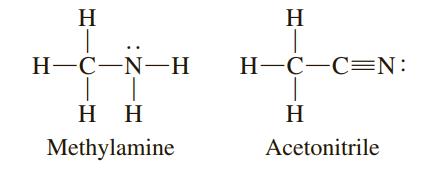
One of the following compounds has a carbon–nitrogen bond length of 116 pm; the other has a carbon–nitrogen bond length of 147 pm. Match a bond length with each compound.
Transcribed Image Text:
H H Н-С—N—Н H-C-C=N: H H H Methylamine Acetonitrile
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Methylamine 147 pm S...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following compounds has a nitrogennitrogen bond: N2, N2H4, N2F2. Match each compound with one of the following bond lengths: 110 pm, 122 pm, 145 pm. Describe the geometry about one of the...
-
Each of the following compounds has three possible names listed for it. For each compound, what is the correct name and why arent the other names used? a. N2O: nitrogen oxide, nitrogen (I) oxide,...
-
The base peak appears at m/z 105 for one of the following compounds and at m/z 119 for the other two. Match the compounds with the appropriate m/z values for their base peaks.
-
In Exercises 1126, determine whether each equation defines y as a function of x. 4x = y 2
-
You are the manager of a stock portfolio worth $10,500,000. It has a beta of 1.15. During the next three months, you expect a correction in the market that will take the market down about 5 percent;...
-
Assuming there are no accidents or delays, the distance that a car travels down an interstate highway can be calculated with the following formula: Distance = Speed Time Create an application that...
-
Depending on the availability of parts, a company can manufacture 3, 4, 5, or 6 units of a certain item per week with corresponding probabilities of 0.10,0.40, 0.30, and 0.20. The probabilities that...
-
Daan Corporation wholesales repair products to equipment manufacturers. On March 1, 2010, Daan Corporation issued $24,000,000 of five-year, 12% bonds at an effective interest rate of 10%, receiving...
-
Cullumber Company started business on January 1, 2024. Some of the events that occurred in its first year of operations follow: Transactions 1. Equipment that cost $208,400 was purchased on February...
-
1. Is it true, as Jack Carter claims, that we cant be accused of being discriminatory because we hire mostly women and minorities anyway? 2. How should Jennifer and her company address the sexual...
-
Which of the following two compounds has the shorter carbonoxygen bond? H :0: - - H Methanol Formaldehyde
-
Calculate the CCl and CC bond lengths in ethyl chloride, C 2 H 5 Cl, using values for the covalent radii from Table 9.4. How do these values compare with the experimental values: CCl, 177; CC, 155...
-
Nutritious Pet Food Companys board of directors declares a cash dividend of $1.00 per common share on November 12. On this date, the company has issued 12,000 shares but 2,000 shares are held as...
-
Use the following information to determine the adjustments, if any, to ICO. (Hint: if you are adjusting ICO, should the adjustments be pre-tax or net of tax?) If you need to increase ICO, enter your...
-
Research 20 solved problems that contains topic of dB Calculation and Nolse Analysis
-
Executives in healthcare organizations consider a number of factors when deciding to pursue a more revolutionary or a more incremental approach to strategy. What are some of those factors, and how...
-
In one hundred words or less, define the theories of Lloyd Bitzer (rhetorical exigence) and Sidney Jourard (self-disclosure) and explain a situation where a rhetorical exigence required that YOU...
-
Your Company enters into a finance lease as the lessee and determines the present value of the lease payments and residual value is $500,000. Your Company also incurs $8,000 in legal fees to execute...
-
Joseph Thompson is president and sole shareholder of Jay Corporation. In December 2013, Joe asks your advice regarding a charitable contribution he plans to have the corporation make to the...
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
A compound of cobalt, carbon, and oxygen contains 28.10% C and 34.47% Co. When 0.147 g of this compound is dissolved in 6.72 g of cyclohexane, the solution freezes at 5.23C. What is the molecular...
-
The carbohydrate digitoxose contains 48.64% carbon and 8.16% hydrogen. The addition of 18.0 g of this compound to 100. g of water gives a solution that has a freezing point of -2.2C. a. What is the...
-
Analysis of a compound gave 39.50% C, 2.21% H, and 58.30% Cl. When 0.855 g of this solid was dissolved in 7.50 g of naphthalene, the solution had a freezing point of 78.0C. The pure solvent freezes...
-
Hi, could someone please help me with this. I need the letter to be nearly two hundred words. This is the assignment below: You are an architect who is working for Dream Homes Company in Toronto....
-
Discussion Board Post - Read the speech example from the textbook below. Then answer the questions in blood at the bottom. "As a veterinarian and owner of a small-animal practice, you work closely...
-
The Essentials of Technical Communication (Tebeaux & Dragga), Oxford U. Press, Third Edition - I. Please explain how a proposal is an argumentative or persuasive piece of writing -- and the four key...

Study smarter with the SolutionInn App


