Calculate the CCl and CC bond lengths in ethyl chloride, C 2 H 5 Cl, using values
Question:
Calculate the C—Cl and C—C bond lengths in ethyl chloride, C2H5Cl, using values for the covalent radii from Table 9.4. How do these values compare with the experimental values: C—Cl, 177; C—C, 155 pm?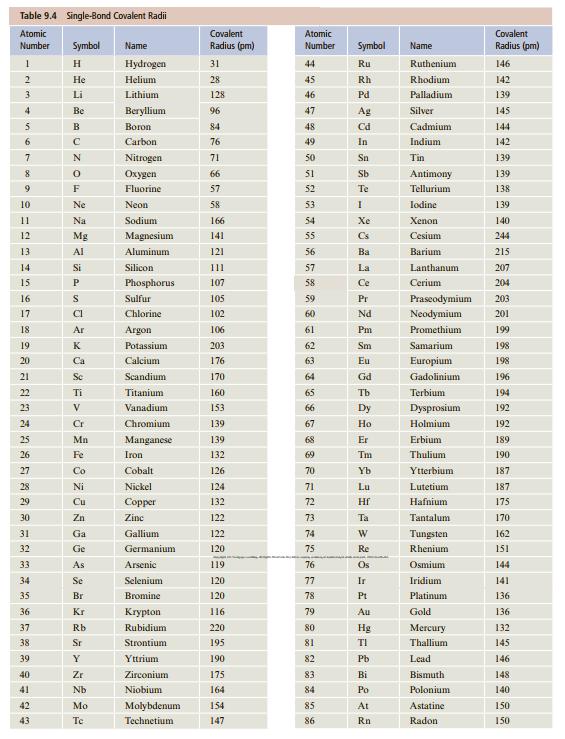
Table 9.4 Single-Bond Covalent Radii Atomic Covalent Atomic Covalent Number Symbol Name Radius (pm) Number Symbol Name Radius (pm) H. Hydrogen 31 44 Ru Ruthenium 146 Не Helium 28 45 Rh Rhodium 142 3 Li Lithium 128 46 Pd Palladium 139 Be Веryllium 96 47 Ag Silver 145 B Вогоn 84 48 Cd Cadmium 144 6. Carbon 76 49 In Indium 142 7 Nitrogen 71 50 Sn Tin 139 8 Охудеn 66 51 Sb Antimony 139 9 F Fluorine 57 52 Te Tellurium 138 10 Ne Neon 58 53 Iodine 139 11 Na Sodium 166 54 Xe Xenon 140 12 Mg Magnesium 141 55 Cs Cesium 244 13 Al Aluminum 121 56 Ba Barium 215 14 Si Silicon 111 57 La Lanthanum 207 15 P. Phosphorus 107 58 Ce Cerium 204 16 Sulfur 105 59 Pr Praseodymium 203 17 CI Chlorine 102 60 Nd Neodymium 201 18 Ar Argon 106 61 Pm Promethium 199 19 K Potassium 203 62 Sm Samarium 198 20 Ca Calcium 176 63 Eu Europium 198 21 Sc Scandium 170 64 Gd Gadolinium 196 22 Ti Titanium 160 65 Tb Terbium 194 23 V Vanadium 153 66 Dy Dysprosium 192 24 Cr Chromium 139 67 Но Holmium 192 25 Mn Manganese 139 68 Er Erbium 189 26 Fe Iron 132 69 Tm Thulium 190 27 Co Cobalt 126 70 Yb Ytterbium 187 28 Ni Nickel 124 71 Lu Lutetium 187 29 Cu Соpper 132 72 Hf Hafnium 175 30 Zn Zine 122 73 Ta Tantalum 170 31 Ga Gallium 122 74 Tungsten 162 32 Ge Germanium 120 75 Re Rhenium 151 33 As Arsenic 119 76 Os Osmium 144 34 Se Selenium 120 77 Ir Iridium 141 35 Br Bromine 120 78 Pt Platinum 136 36 Kr Кгуpton 116 79 Au Gold 136 37 Rb Rubidium 220 80 Hg Mercury 132 38 Sr Strontium 195 81 TI Thallium 145 39 Y Yttrium 190 82 Pb Lead 146 40 Zr Zirconium 175 83 Bi Bismuth 148 41 Nb Niobium 164 84 Po Polonium 140 42 Mo Molybdenum 14 85 At Astatine 150 43 Te Technetium 147 86 Rn Radon 150
Step by Step Answer:
d CCl r C r Cl 76 pm 102 pm 178 pm ...View the full answer



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
Calculate the CH and CCl bond lengths in chloroform, CHCl3, using values for the covalent radii from Table 9.4. How do these values compare with the experimental values: CH, 107 pm; CCl, 177 pm?...
-
The following table shows the UKs Retail Price Index from 1970 to 2009. As you can see, the index was reset to 100 in January 1974, and again in January 1987. What is the annual rate of inflation for...
-
The financial statements for Quebecor Inc. and Thomson Reuters Corporation may be found on SEDAR (www.sedar.com) or the companies' websites. Instructions (a) 'What business is Quebecor Inc. in? Is...
-
The graph of f(x) = ax 2 + bx + c is shown in the figure. Solve each inequality. (a) f(x) < 0 (b) f(x) = 0 I 3 -y=f(x)
-
You are the manager of a bond portfolio of $10 million face value of bonds worth $9,448,456. The portfolio has a yield of 12.25 percent and a duration of 8.33. You plan to liquidate the portfolio in...
-
A 15-year annuity pays $1,475 per month, and payments are made at the end of each month. If the interest rate is 9 percent compounded monthly for the first seven years, and 6 percent compounded...
-
Financial statements of iPud Ltd are presented below. Additional information 1. Payables includes $5620 (2025) and $5730 (2023) trade accounts payable; the remainder is accrued expenses. Market...
-
A mixture of n-hexane vapor and air leaves a solvent recovery unit and flows through a 70-cm diameter duct at a velocity of 3.00 m/s. At a sampling point in the duct the temperature is 40C, the...
-
1. What are some elements of a code of ethics for criminology/criminal justice research? The code of ethics sets up the rules and the principles of an organization that leads behavior. It could be...
-
Three former RIM employees decided to go into business for themselves and open a store near an office park to sell wireless equipment to young professionals. Their first products were cell phones,...
-
One of the following compounds has a carbonnitrogen bond length of 116 pm; the other has a carbonnitrogen bond length of 147 pm. Match a bond length with each compound. H H -N H-C-C=N: H H H...
-
What do you expect for the BCl bond length in boron trichloride, BCl 3 , on the basis of covalent radii (Table 9.4)? Table 9.4 Single-Bond Covalent Radii Atomic Covalent Atomic Covalent Number Symbol...
-
Assume a company with the following balance sheet accounts: You are concerned only about overstatements of owner's equity. Set tolerable misstatement for the three relevant accounts such that the...
-
The Government of Canada recently signed new preferential trade agreement, and has agreed to pay subsidies to turkey farmers to compensate for increased competition they may face from imports. You...
-
K Find f'(-3) if f(x)= +3 - 10x. 6 f'(-3)= (Simplify your answer. Type an integer or a fraction.)
-
How can mediation and arbitration be used to resolve conflicts, and what are their advantages and limitations compared to direct negotiations?
-
Find the volume of a pyramid with a height of 2 0 cm and a rectangular base with dimensions of 4 cm and 9 cm .
-
Find the periodic payment R required to amortize a loan of P dollars over t years with interest charged at the rate of r%/year compounded m times a year. (Round your answer to the nearest cent.) P =...
-
Develop a plan for an event (e.g., golf tournament; gala ball; annual meeting; or a series of lectures, workshops, or seminars where a fee is charged for attendance or participation) for your...
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
Fish blood has an osmotic pressure equal to that of seawater. If seawater freezes at 2.3C, what is the osmotic pressure of the blood at 25C? To solve this problem, what assumptions must be made?
-
Four ways were discussed to express the concentration of a solute in solution. Identify them and define each concentration unit.
-
A salt solution has an osmotic pressure of 17 atmospheres at 22C. What is the freezing point of this solution? What assumptions must be made to solve this problem?
-
ow do synergistic dynamics emerge within high-performance teams, and what role do diverse skill sets, complementary roles, and shared goals play in fostering collaborative innovation and collective...
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
You will complete two case studies over the course of the semester. These are to be completed individually. Each case study is found in the textbook and there are questions there to help guide your...

Study smarter with the SolutionInn App


