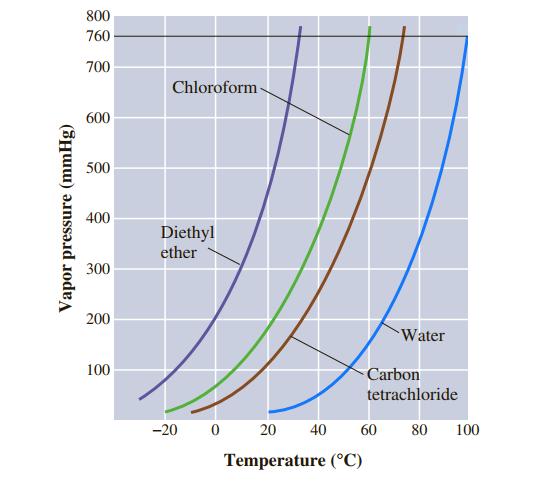
Use Figure 11.7 to estimate the boiling point of carbon tetrachloride, CCl 4, under an external pressure
Question:
Use Figure 11.7 to estimate the boiling point of carbon tetrachloride, CCl4, under an external pressure of 250 mmHg.
Transcribed Image Text:
800 760 700 Chloroform- 600 500 400 Diethyl ether 300 200 Water 100 Carbon tetrachloride -20 20 40 60 80 100 Temperature (°C) Vapor pressure (mmHg)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
Dropping a line from the inter...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use Figure 11.7 to estimate the boiling point of diethyl ether, (C 2 H 5 ) 2 O, under an external pressure of 470 mmHg. 800 760 700 Chloroform- 600 500 400 Diethyl ether 300 200 Water 100 Carbon...
-
Using the vapor-pressure curves in Figure 11.25, (a) Estimate the boiling point of ethanol at an external pressure of 200 torr; (b) Estimate the external pressure at which ethanol will boil at 60 oC;...
-
Use Figure 10.8 to estimate the boiling point of ethanol at 400 torr.
-
a. Find the nth-order Taylor polynomials for the given function centered at the given point a, for n = 0, 1, and 2.b. Graph the Taylor polynomials and the function. f(x) = ln x, a e =
-
Suppose FRM, Inc., issued a zero coupon, equity index-linked note with a five-year maturity. The par value is $1,000, and the coupon payment is stated as 75 percent of the equity index return or as...
-
Lamplighter Company, the lessor, agrees to lease equipment to Tilson Company, the lessee, beginning January 1, 2019. The lease terms, provisions, and related events are as follows: The lease is...
-
You draw four cards, one at a time, from a standard deck. You note the suit and replace the card in the deck. The random variable represents the number of cards that are diamonds. Determine whether...
-
Consider the following data, which relate to the two divisions of McIntyre Products. Required Compare the two divisions in terms of return on investment and residual income. In the past year, which...
-
Two resistors are connected in parallel with an 99 volt battery. The resistors have values of R 1 = 7 ohms and R 2 = 55 ohms. What is the current (in amps) in this circuit? Round your answer to two...
-
You are given a series of trades. Each trade has a key, a value, a quantity, and a sequence number. For each trade, print the weighted moving average of all the trades for that particular key. Round...
-
Chloroform, CHCl 3 , a volatile liquid, was once used as an anesthetic but has been replaced by safer compounds. Chloroform boils at 61.7C and has a heat of vaporization of 31.4 kJ/mol. What is its...
-
An element crystallizes with a simple cubic lattice with atoms at all the lattice points. If the radius of the atom is 200. pm, what is the volume of the unit cell? a. 8.00 10 6 pm 3 b. 6.40 10 7...
-
Refer to the data for AnimPix, Inc., in Problem 8-26. In addition, the company has provided the following details concerning its activity rates: Management has provided the following ease of...
-
Determine the heat rejected in kJ / kg to a simple ideal steam Rankine cycle with a condenser pressure of 2 0 kPa and a steam generator with a pressure of 2 MPa. ( The steam leaves the steam...
-
7. A rocket of a mass 200 kg loaded with a propellant of a mass 1200 kg is powered by a rocket engine whose exhaust velocity is 5000 kmph. What is the maximal fuel consumption per second possible,...
-
parallel, perpendicular, or neither. line py=x+2 line qy-2x+1 state whether line p and line qare Sime po passes through (4,-2) and (3,9) line qy-11x + 5 line p is parallel to y 3r-8 line q is...
-
Please provide a short paragraph response for each of the questions below. Do you think it's right or justified to have an unregulated Marketing Research industry? How do you see the role of...
-
What do you believe are the barriers to successful communication in the world of work and what would you recommend overcoming them?
-
List the four components of Gross Domestic Product(GDP) and provide an example of each. Explain how each item affects you and the way that you live today
-
(a) What is the focal length of a magnifying glass that gives an angular magnification of 8.0 when the image is at infinity? (b) How far must the object be from the lens?
-
The bond length in C2 is 131 pm. Compare this with the bond lengths in C2H2 (120 pm), C2H4 (134 pm), and C2H6 (153 pm). What bond order would you predict for C2 from its bond length? Does this agree...
-
Draw resonance formulas of the nitric acid molecule, HNO3. What is the geometry about the N atom? What is the hybridization on N? Use bond energies and one Lewis formula for HNO3 to estimate Hf for...
-
One resonance formula of benzene, C6H6, is What is the other resonance formula? What is the geometry about a carbon atom? What hybridization would be used in valence bond theory to describe the...
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


