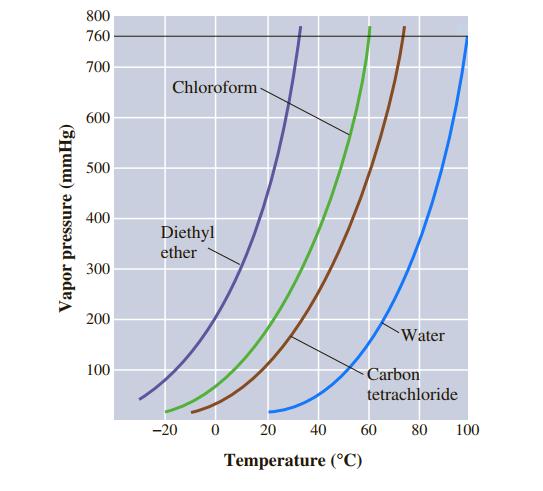
Use Figure 11.7 to estimate the boiling point of diethyl ether, (C 2 H 5 ) 2
Question:
Use Figure 11.7 to estimate the boiling point of diethyl ether, (C2H5)2O, under an external pressure of 470 mmHg.
Transcribed Image Text:
800 760 700 Chloroform- 600 500 400 Diethyl ether 300 200 Water 100 Carbon tetrachloride -20 20 40 60 80 100 Temperature (°C) Vapor pressure (mmHg)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
Dropping a line from the int...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the vapor-pressure curves in Figure 11.25, (a) Estimate the boiling point of ethanol at an external pressure of 200 torr; (b) Estimate the external pressure at which ethanol will boil at 60 oC;...
-
Use Figure 10.8 to estimate the boiling point of ethanol at 400 torr.
-
Use Figure 11.7 to estimate the boiling point of carbon tetrachloride, CCl 4, under an external pressure of 250 mmHg. 800 760 700 Chloroform- 600 500 400 Diethyl ether 300 200 Water 100 Carbon...
-
a. Find the nth-order Taylor polynomials for the given function centered at the given point a, for n = 0, 1, and 2.b. Graph the Taylor polynomials and the function. f(x)=x, a = 9
-
An investment manager expects a stock to be quite volatile and is considering the purchase of either a straddle or a chooser option. The stock is priced at 44, the exercise price is 40, the...
-
The following beginning balance sheet and statement of cash flows for 2019 are available for Fazzi Company: Required: On the basis of this information, prepare a balance sheet for Fazzi as of...
-
A person is required to toss 8 unbiased coins and note down the outcome of each. The random variable represents the number of heads. Determine whether the experiment is a binomial experiment. If it...
-
William H. Bailey, MD, executed a note payable to California Dreamstreet, a joint venture that solicited investments for a cattle breeding operation. Baileys promissory note read, Dr. William H....
-
Blood flows through the aorta at an average speed of v = 19cm/s. The aorta is roughly cylindrical with a radius r = 17.6 mm. The volume rate of blood flow through the aorta is r 2 v . Calculate the...
-
Walton Ltd. is considering replacing an existing machine with a new and faster machine that will produce a more reliable product (i.e., better tolerances). The switch to a new machine resulting in a...
-
Chloroform, CHCl 3 , a volatile liquid, was once used as an anesthetic but has been replaced by safer compounds. Chloroform boils at 61.7C and has a heat of vaporization of 31.4 kJ/mol. What is its...
-
An element crystallizes with a simple cubic lattice with atoms at all the lattice points. If the radius of the atom is 200. pm, what is the volume of the unit cell? a. 8.00 10 6 pm 3 b. 6.40 10 7...
-
Ruth Ames died on January 10, 2021. In filing the estate tax return, her executor, Melvin Sims, elects the primary valuation date and amount (fair market value on the date of death). On March 12,...
-
Patterson Development sometimes sells property on an installment basis. In those cases, Patterson reports income in its income statement in the year of the sale but reports installment income by the...
-
The president was the principal promoter and president of a corporation. He paid for the shares he had subscribed for. The corporation became insolvent and made an assignment for the benefit of its...
-
Question Content Area Net Present Value Carsen Sorensen, controller of Thayn Company, just received the following data associated with production of a new product: Expected annual revenues: $740,000...
-
8. An airplane is flying N75E with an airspeed of 550 mph and a 45 mph wind blowing S75E. What is the actual speed, in mph and direction, written as a bearing, of the plane? Round to the nearest 1...
-
Krystal suggests that the tax code allowing for a stepped - up basis ( a code section that allows heirs to receive inherited property with a basis that equals FMV at the time of death ) is flawed and...
-
The concept of GDP is used by policy makers for many purposes that include keeping track of the growth of our economy and forecasting a recession; but it is also abused by some. Discuss some...
-
On October 31 Juanita Ortega, owner of Outback Guide Service, received a bank statement dated October 30. Juanita found the following: 1. The checkbook has a balance of $2,551.34. 2. The bank...
-
What are the differences between a bonding and an antibonding molecular orbital of a diatomic molecule?
-
Describe the formation of molecular orbitals resulting from the interaction of two 2p orbitals.
-
Describe the bonding in O3, using molecular orbital theory. Compare this with the resonance description.
-
You are planning to retire in 30 years. You want to be able to spend $40,000 per year in retirement, adjusted for inflation (so you will spend the equivalent in each year of $40,000 in today's...
-
From a lawsuit, you have been awarded a 31-payment, constant growth annuity. The first payment is at Year O and is equal to $380, and each subsequent payment will be paid in 16 month intervals, with...
-
You will receive 13 payments of $535, where the first payment will be received today (Month 0) and all other payments will be received in 10-month intervals (Months 10, 20, 30 ... 120). Assume that...

Study smarter with the SolutionInn App


