Use the solubility rules (Table 4.1) to decide which of the following compounds are expected to be
Question:
Use the solubility rules (Table 4.1) to decide which of the following compounds are expected to be soluble and which insoluble.
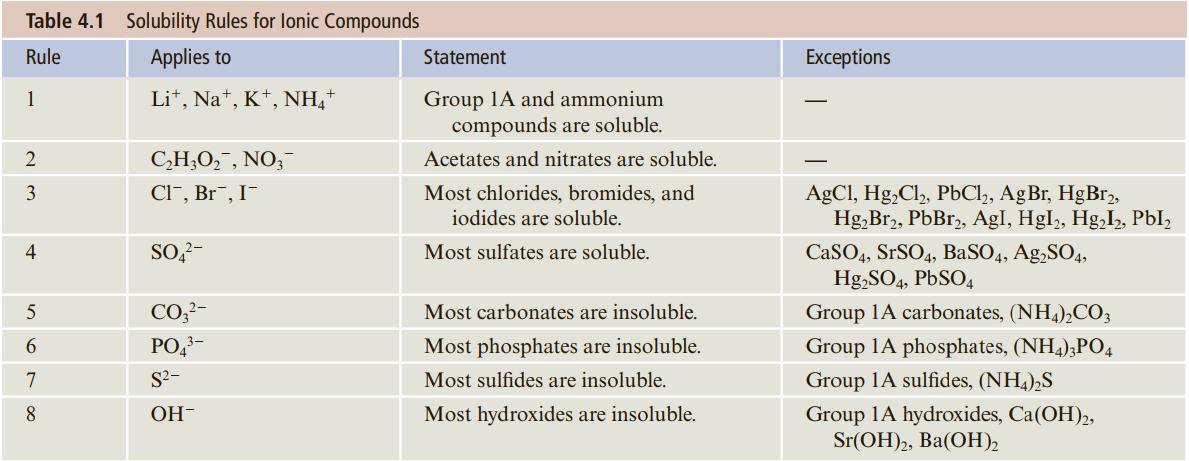
a. Mg(C2H3O2)2
b. NiS
c. Cr(NO3)2
d. Ca3(PO4)2
Transcribed Image Text:
Table 4.1 Solubility Rules for lonic Compounds Rule Applies to Statement Exceptions Lit, Na*, K*, NH," Group 1A and ammonium compounds are soluble. 1 C,H;O,, NO,- Acetates and nitrates are soluble. 3 CI", Br", I- Most chlorides, bromides, and AgCl, Hg,Cl,, PbCl,, AgBr, HgBr, Hg,Br2, PbBr,, AgI, Hgl, Hg,I, PbI, iodides are soluble. SO,?- CaSO4, SrSO4, BaSO4, Ag,SO4, Hg,SO4, PBSO4 4 Most sulfates are soluble. 5 CO,?- Most carbonates are insoluble. Group 1A carbonates, (NH4),CO; 6 PO,- Most phosphates are insoluble. Group 1A phosphates, (NH4),PO4 7 S2- Most sulfides are insoluble. Group 1A sulfides, (NH,),S 8 OH- Most hydroxides are insoluble. Group 1A hydroxides, Ca(OH),, Sr(ОH)2, Ba(ОН).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a MgC 2 H 3 O 2 2 is soluble acetates are solub...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds are aromatic? a. b. c. Cycloheptatrienyl cation d. e. f. g. Cyclononatetraenyl anion h. CH2=CHCH=CHCH=CH2
-
Which of the following compounds are chiral? (a) 2-Methylheptane (b) 3-Methylheptane (c) 4-Methylheptane (d) 1,1-Dibromopropane (e) 1,2-Dibromopropane (f) 1,3-Dibromopropane (g) Ethene, H2C=CH2 (h)...
-
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may...
-
Find the minimum and maximum values of the function subject to the given constraint. f(x, y) =xy, 4x +9y = 32
-
The study of whether birth decade can predict the number of software millionaires born in the decade. The data are reproduced in the table shown below. a. Construct a 95% confidence interval for the...
-
Tyneka inherited 1,000 shares of Aqua, Inc. stock from Joe. Joes basis was $35,000, and the fair market value on July 1, 2019 (the date of death) was $45,000. The shares were distributed to Tyneka on...
-
A multicomponent mixture is boiled in a flask at \(1 \mathrm{~atm}\). The vapors are condensed and recovered as a liquid product. It is desired to examine the mole fractions of the residual liquid in...
-
The total processing cost for producing the X-Pacer running shoe in Problem 2-5 is $18. The Omega Shoe Company starts production of 650 pairs of the shoes weekly, and the average weekly yield is 90%,...
-
Applying analytical synthesis of mechanisms, design a four-bar mechanism to move the object shown in the figure, through the three positions indicated in their numbered order without taking into...
-
As the owner of Barney??s Broilers??a fast-food chain??you see an increase in the demand for broiled chicken as consumers become more health conscious and reduce their consumption of beef and fried...
-
You are given a solution of the ions Mg 2+ , Ca 2+ , and Ba 2+ . Devise a scheme to separate these ions using sodium sulfate. Note that magnesium sulfate is soluble.
-
The solubility of cobalt(II) iodate in water is 1.2 g/100 mL. Calculate the solubility product constant for cobalt(II) iodate, Co(IO 3 ) 2 .
-
Using Figure 6.4 as a model, illustrate the operation of HEAPSORT on the array A = 5, 13, 2, 25, 7, 17, 20, 8, 4.
-
The Cool Watch company sells two products. The company's water-resistant model sells for $100, has a contribution margin of 35%, and generated sales of $100,000. The company's "basic timepiece" sells...
-
A 8 . 2 2 - nC charge is located 1 . 6 3 m from a 4 . 2 6 - nC point charge. ( a ) Find the magnitude of the electrostatic force that one charge exerts on the other.
-
A garden hose can fill a swimming pool in 9 days, and a larger hose can fill the pool in 4 days. How long will it take to fill the pool if both hoses are used?
-
A car is making a trip in a total time of 1 0 4 min. During the first 3 7 min its average speed is 2 8 . 5 mph. What average speed of the rest of the trip is needed if the car\'s overall average...
-
5. The burning of organic compounds involves a chemical reaction with oxygen from the air. This reaction is called a combustion reaction. Most organic compounds react with oxygen to produce carbon...
-
What is the resonant frequency of a 0.500 mH inductor connected to a 40.0 F capacitor?
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Carbon monoxide and hydrogen react in the presence of a catalyst to form methanol, CH3OH: An equilibrium mixture of these three substances is suddenly compressed so that the concentrations of all...
-
A and B react to produce C according to the following chemical equation: A+ B C Amounts of A and B are added to an equilibrium reaction mixture of A, B, and C such that when equilibrium is again...
-
Given the hypothetical exothermic reaction A2(g) + 2B(g) 2AB(g) at equilibrium, decide which of the following containers represents the reaction mixture at the higher temperature? (The other...
-
ow do synergistic dynamics emerge within high-performance teams, and what role do diverse skill sets, complementary roles, and shared goals play in fostering collaborative innovation and collective...
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
You will complete two case studies over the course of the semester. These are to be completed individually. Each case study is found in the textbook and there are questions there to help guide your...

Study smarter with the SolutionInn App


