Using the ionic radii given in Table 9.3, estimate the energy to form a mole of Na
Question:
Using the ionic radii given in Table 9.3, estimate the energy to form a mole of Na+F- ion pairs from the corresponding atomic ions.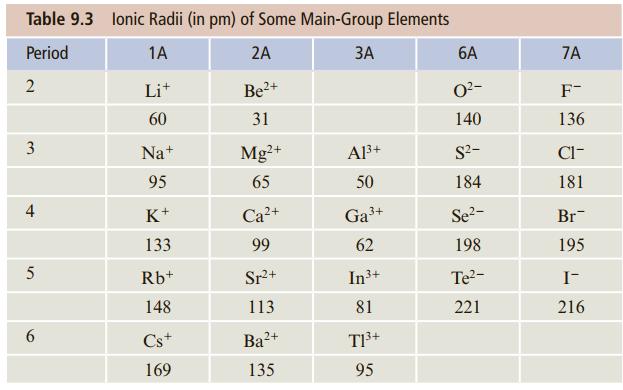
Transcribed Image Text:
Table 9.3 lonic Radii (in pm) of Some Main-Group Elements Period 1A 2A ЗА 6A 7A 2 Lit 2+ O?- F- 60 31 140 136 3 Na+ Mg²+ Al3+ S?- Cl- 95 65 50 184 181 4 K+ Са+ Ga3+ Se2- Br- 133 99 62 198 195 Rb+ Sr+ In3+ Te?- I- 148 113 81 221 216 6. Cs+ Ba?+ T3+ 169 135 95
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
r Na 95 pm r F 136 pm d ion pair r Na r F 95 pm 136 pm ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From the ionic radii given in Figure 7.7, calculate the potential energy of a Ca2+ and O2- ion pair that is just touching (the magnitude of the electronic charge is given on the back inside cover)....
-
From Equation 8.4 and the ionic radii given in Figure 7.7, calculate the potential energy of the following pairs of ions. Assume that the ions are separated by a distance equal to the sum of their...
-
In the ionic compounds LiF, NaCl, KBr, and RbI, the measured cation-anion distances are 2.01 Ã (Li-F), 2.82 Ã (Na-Cl), 3.30 Ã (K-Br), and 3.67 Ã (Rb-I), respectively. (a)...
-
In Exercises 118, find the distance between each pair of points. If necessary, express answers in simplified radical form and then round to two decimals places. (2.6, 1.3) and (1.6, -5.7)
-
On January 2 of a particular year, an American firm decided to close out its account at a Canadian bank on February 28. The firm is expected to have 5 million Canadian dollars in the account at the...
-
The following transactions were selected from the records of Evergreen Company: July 12 Sold merchandise to Wally Butler, who paid the $1,000 purchase with cash. The goods cost Evergreen Company...
-
Information from the records of Smart Systems Pty Ltd for the year ended 30 June 2025 is given below. Required Calculate the cost of work in process inventory on 1 July 2024. Factory overhead, 200%...
-
Bel-Air, Inc., is a small engineering corporation that surveys land for development. The company has grown rapidly over the past few years, and management has to decide whether to hire new engineers...
-
Letty, Brian, Han, Dominic, and Gisele loved driving around town fast and furious. If one happened to be speeding and noticed an officer or a state trooper, they would send signals to others to make...
-
Two forces P and Q are applied to the lid of a storage bin as shown. Knowing that P = 60 N and Q = 48 N, determine by trigonometry the magnitude and direction of the resultant of the two forces. %3D...
-
Calculate the difference in electronegativities between the atoms in SrF 2 and between the atoms in SnF 2 . Which substance would you expect to be more ionic in character? One of these compounds...
-
An ion M 2+ has the configuration [Ar]3d 2 , and an atom has the configuration [Ar]4s 2 . Identify the ion and the atom.
-
Briefly summarize the elements of ISO 9001.
-
Serum iron was measure in 60 healthy adults. The sample mean of serum iron was 18.30 with a sample standard deviation of 4.66. Based on the date estimate the population mean serum level in healthy...
-
The company Amazon acquired raw materials whose cash value is $586,250.90 on credit, with a down payment of 30% on the cash price, and the balance through advance monthly payments of $15,250.00 and a...
-
Case Study Reflective: this makes scents PURPOSE: The purpose of this reflection is to think about the information provided in the case study and how you will integrate your learnings in the working...
-
How much Alberta must deposit to accumulate $250,000 within 4 years at an interest rate of 8%: a) Capitalized monthly b) Capitalized quarterly
-
Tamar Company manufactures a single product in two departments: Forming and Assembly. Information for the Forming process for May follows. Direct Materials Conversion Units Percent Complete Percent...
-
If households and firms were the only two sectors in economy, income and output would be equal...True or false
-
For Problem estimate the change in y for the given change in x. y = f(x), f'(12) = 30, x increases from 12 to 12.2
-
Snow forms in the upper atmosphere in a cold air mass that is supersaturated with water vapor. When the snow later falls through a lower, warm air mass, rain forms. When this rain falls on a sunny...
-
The percent relative humidity of a sample of air is found as follows: (partial pressure of water vapor/vapor pressure of water) 100. A sample of air at 21C was cooled to 15C, where moisture began to...
-
A sample of air at 21C has a relative humidity of 58%. At what temperature will water begin to condense as dew? (See Problem 11.97.) Problem 11.97 The percent relative humidity of a sample of air is...
-
A company is faced with the decision of whether to lease or purchase new equipment. The equipment can be leased at an annual cost of $210,000 or purchased for $560,000. The equipment has an expected...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...
-
A stock had returns of 28%, 1%, and 13% over the previous 3 years. The average return on this stock over the 3-year period was 14%. What was the variance of this stock's returns over the previous 3...

Study smarter with the SolutionInn App


