What is G at 25C for the reaction Sn 2+ (aq) + 2Hg 2+ (aq) Sn
Question:
What is ∆G° at 25°C for the reaction
Sn2+(aq) + 2Hg2+(aq) → Sn4+(aq) + Hg22+(aq)
For data, see Table 19.1.
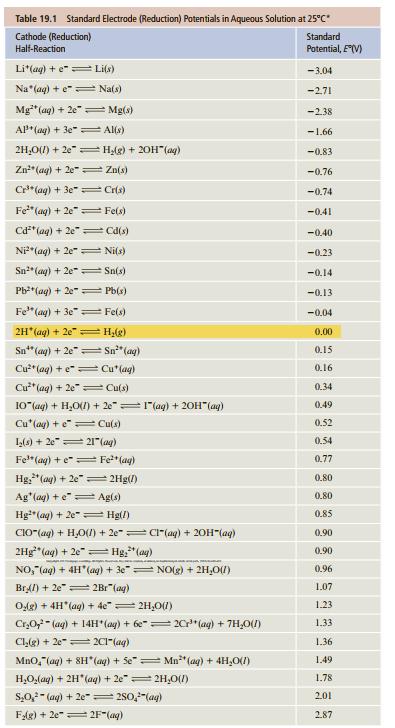
Transcribed Image Text:
Table 19.1 Standard Electrode (Reduction) Potentials in Aqueous Solution at 25°C Standard Cathode (Reduction) Half-Reaction Potential, E(V) Li*(ag) + e"= Li(s) - 3.04 Na"(aq) + e"= Na(s) -2.71 Mg"(ag) + 2e" = Mg(s) -2.38 AP* (ag) + 3e" = Al(s) -1.66 2H,O(1) + 2e"= Hlg) + 20H"(ag) -0.83 Zn"(ag) + 2e = Zn(s) -0.76 Cr*(aq) + 3e" Cris) -0.74 Fe" (ag) + 2e" Fe(s) -0.41 Cd*(ag) + 2e" = Cd(s) -0.40 Ni*(aq) + 2e" = Ni(s) -0.23 Sn"(ag) + 2e Sn(s) -0.14 Pb*(ag) + 2e" = Pb(s) -0.13 Fe" (ag) + 3e" = Fe(s) -0.04 2H (ag) + 2e H,(g) 0.00 Sn" (ag) + 2e"= Sn" (ag) 0.15 Cu**(aq) + e"= Cu*(ag) Cu**(ag) + 2e"= Cu(s) 10"(ag) + H,O) + 20"=1"(ag) + 20H" (ag) 0.16 0.34 0.49 Cu'(ag) + e= Cu(s) 0.52 0.54 L(s) + 2e"= 21"(ag) Fe"(ag) + e= Fe*(ag) 0.77 Hg;" (ag) + 2e =2Hg(/) 0.80 Ag'(ag) + e" Ag(s) 0.80 Hg"(ag) + 2e" = Hg(/) 0.85 CIO"(ag) + H,O() + 2e"= CI"(aq) + 20H"(ag) 2Hg" (aq) + 2e" = Hg," (ag) NO," (ag) + 4H (ag) + 3e"= NO(g) + 2H,01) 0.90 0.90 0.96 Brl) + 2e" 2Br"(ag) 1.07 Odg) + 4H"(ag) + 4e"= 2H,0(7) 1.23 Cr,O,- (ag) + 14H (ag) + 6e"= 2C"(ag) + 7H,0(1) 1.33 Cl,g) + 2e"= 2C"(ag) 1.36 Mno,"(aq) + SH (aq) + Se" Mn** (ag) + 4H,0(1) 1.49 H,O(ag) + 2H"(aq) + 2e"= 2H,0(1) 1.78 S,0,- (ag) + 2e-=280,"(ag) 2.01 FAg) + 2e" 2F"(ag) 287
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The halfcell reactions the corresponding halfcell potentials and their sums are displayed ...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Table 10.16 gives data on the crime rate in 47 states in the United States for 1960. Try to develop a suitable model to explain the crime rate in relation to the 14 socioeconomic variables given in...
-
Attached are two data sets, one concerning Skittles and one about cereals. You may assume both samples provided were selected randomly and are representative of the population. Be sure to use the...
-
In Example 15.6, pure-component, liquid-phase adsorption data are used with the extended-Langmuir isotherm to predict a binary-solute data point. Use the following mixture data to obtain the best fit...
-
Find the volumes of the solids in Problems 4952 correct to the nearest unit. 4 cm -6 cm 8 cm
-
One of the most difficult tasks of developing and managing a global portfolio is assessing the risks of potential foreign investments. Duke University researcher C. R. Henry collaborated with two...
-
creative, innovative, or particularly effective facility management practice. Describe your example - what is the purpose? What makes it creative, innovative, or particularly effective? What would it...
-
As discussed in Example 6.7, toluene \(\left(\mathrm{C}_{7} \mathrm{H}_{8} ight)\) is to be converted thermally to benzene \(\left(\mathrm{C}_{6} \mathrm{H}_{6} ight)\) in a hydrodealkylation...
-
This financial information is available for Klinger Corporation. The weighted-average number of shares of common stock outstanding was 180,000 for 2013 and 150,000 for 2014. Instructions Calculate...
-
Pelcher Company manufactures soccer balls in two sequential processes: Cutting and Stitching. All direct materials enter production at the beginning of the Cutting process. The following Information...
-
Supreme Videos, Inc., produces short musical videos for sale to retail outlets. The company?s balance sheet accounts as of January 1, the beginning of its fiscal year, are given on the following...
-
Define the faraday.
-
Consider the reducing agents Sn 2+ (aq), Cl 2 (g), and I (aq). Which is strongest? Which is weakest?
-
M. Jordan & E. Stone, CPAs, audited the financial statements of Tech Company, a non issuer, for the year ended December 31, 2016, and expressed an unmodified opinion. For the next year, ended...
-
One year ago, Jack and Jill set up a vinegarbottling firm (called JJVB). Use the following data to calculate JJVBs opportunity cost of production during its first year of operation: Jack and Jill...
-
Illustrate the connection between Sues AP, MP, AVC , and MC curves in graphs like Fig. 11. 7 . Fig. 11. 7 Sues Surfboards, in Problem 2 , hires workers at $500 a week and its total fixed cost is...
-
Hanjin to Shut Down European Operations Following a prolonged slump in the shipping market, Hanjin Shipping, a South Korean logistics and container transport firm, scaled down its operations by...
-
In 2020, Toni taught music and earned $20,000. She rented out her basement and earned $4,000. On January 1, 2021, she quit teaching, stopped renting out her basement, and began to use it as the...
-
a. Why do you think Huawei worked with other firms to produce the P9 smartphone rather than performing all the required tasks itself ? b. What are the roles of transactions costs, economies of scale,...
-
(a) To what temperature must you raise a resistor made of constantan to double its resistance, assuming a constant temperature coefficient of resistivity? (b) To cut it in half? (c) What is...
-
You work as an operations consultant for a textile company. Your client has a well-established distribution system in the US market. The company has hundreds of stores and four distribution centers....
-
Formic acid, HCHO2, is a stronger acid than acetic acid, HC2H3O2. Which is the stronger base, formate ion, CHO2, or acetate ion, C2H3O2?
-
Rank the following solutions from most acidic to most basic (water molecules have been omitted for clarity).
-
You have solutions of NH3, HCl, NaOH, and HC2H3O2 (acetic acid), all with the same solute concentrations. Rank these solutions in order of pH, from the highest to the lowest.
-
An automobile cooling system holds 16 L of water. How much heat does it absorb if its temperature rises from 20 C to 80 C? A 5.0 kg lead ball is dropped into a 2.0 L insulated pail of water initially...
-
A 15.0 kg box is allowed to slide from rest down a ramp that makes an angle of 30.0 o to the horizontal. The coefficient of friction between the box and the ramp is 0.400 and the ramp is 5.00 m long....
-
1. A bowling ball rolls without slipping up a ramp that slopes upward at an angle beta to the horizontal. Treat the ball as a uniform, solid sphere, ignoring the finger holes. What is the...

Study smarter with the SolutionInn App


