In Example 15.6, pure-component, liquid-phase adsorption data are used with the extended-Langmuir isotherm to predict a binary-solute
Question:
In Example 15.6, pure-component, liquid-phase adsorption data are used with the extended-Langmuir isotherm to predict a binary-solute data point. Use the following mixture data to obtain the best fit to an extended Langmuir-Freundlich isotherm of the form
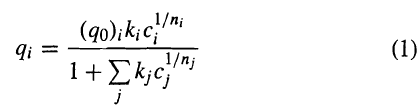
Data for binary-mixture adsorption on activated carbon (1000 m2/g) at 25°C for acetone (1) and propionitrile (2) are asfollows:
Transcribed Image Text:
1/n¡ (qo);k;c" qi 1+Σg (1) 1/nj
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
From Eq 6 in Example 156 the extended LangmuirFreundlich equations are Fit mixture adsorp...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
The adsorption of a gas is described by the Langmuir isotherm with K = 0.777 kPa-1 at 25C. Calculate the pressure at which the fractional surface coverage is (a) 0.20, (b) 0.75.
-
The excess Gibbs energy of u binary liquid mixture at T and P is given by: GR/RT = (- 2.6x1 - l.8x2)x1x2 Find expressions for In y1 and In y2 at T and P. (a) Show that when these expressions are...
-
In Example 6 we used data from 1920 to 2020 and found that the life span in the United States depended on the year of birth according to the equation L(x) = 11.027 + 14.304 ln x where x is the number...
-
Why does allocating an array of length \(n\) take time proportional to \(n\) ?
-
Describe the effects damage estimates would have on the financial statements of a corporation and a partnership? How do disclosure requirements differ from a corporation to a partnership and what...
-
Scheduled debt payments of $600 each are due three months and six months from now. If interest at 10% is allowed, what single payment today is required to settle the two scheduled payments?
-
On November 14, 2010, Amazing Sound, Inc., sold $3,100 of inventory (cost is $1,330) on account to one of its customers. The terms were 3/10, n/30, FOB desti nation. On November 16, Amazing Sound,...
-
The following elements of stockholders equity are adapted from the balance sheet of Sandler Marketing, Corp. Sandler paid no preferred dividends in 2011. Requirement 1. Compute the dividends to the...
-
What role do cultural embeddedness and institutionalization play in shaping the resilience and longevity of organizational culture, and how can organizations strategically leverage cultural dynamics...
-
The dividends are at $1.00 and $1.15, respectively. As financial reporting advisors, we are asked by the head of Sterling Company to advise in accounting for stock investments. This is the first year...
-
Lewis, Gilliland, Chertow, and Hoffman [J. Am. Chem. Soc., 72, 1153-1 157 (1950)] measured adsorption equilibria for pure propane, pure propylene, and binary mixtures thereof, on activated carbon and...
-
Sircar and Myers [J. Phys. Chem., 74,2828-2835 (1970)] measured liquid-phase adsorption at 30?C for a binary mixture of cyclohexane (1) and ethyl alcohol (2) on activated carbon. Assuming no...
-
On October 1, 2019, Wenke Corp signs a lease to use a piece of equipment for seven years. The lease requires Wenke to make seven payments of $27,865 the first of which is on October 1, 2019, the date...
-
A craft coffee shop located in the lobby of a downtown skyscraper sells a cup of coffee for $ 6 . 0 0 . Employees who work in a building three blocks away have an easy access to a food truck that...
-
Generate a half page- one page report for each topic about Dutch Bros Page 1: Brand awareness, brand association, and brand personality Page 2: Competition Analysis Page 3: Current Marketing...
-
An arc-welding operation on nickel performs a groove weld, whose cross-sectional area = 29 mm 2 . Travel velocity = 3.9 mm/s. Heat transfer factor = 0.83 and melting factor = 0.64. Determine the rate...
-
In project management, organisational structure plays a vital role in determining how projects are planned, executed, and controlled. Unlike traditional hierarchical structures, the matrix structure...
-
Relate the Gaps model to some of the examples from the session 1 discussion. Which gaps (or lack thereof) contributed to either good or bad service experiences? Which gaps are the most or least...
-
If circulation of water takes place by convection currents, set up during the heating of water, the boiler is known as: (a) Natural circulation boiler (b) Forced circulation boiler (c) Internally...
-
Floyd Distributors, Inc., provides a variety of auto parts to small local garages. Floyd purchases parts from manufacturers according to the EOQ model and then ships the parts from a regional...
-
On March 2, 2000, 3Com, a profitable provider of computer networking products and services, sold 5 percent of one of its subsidiaries, Palm, to the public via an initial public offering (IPO). 3Com...
-
Nitrogen gas initially at 8.5 bar expands isentropically to 1 bar and 150 C Assuming nitrogen to be an ideal gas, calculate the initial temperature and the work produced per mole of nitrogen.
-
Combustion products from a burner enter a gas turbine at 10 bar and 950oC and discharge at 1.5 bar. The turbine operates adiabatically with an efficiency of 775K assuming the combustion products to...
-
Isobutane expands adiabatically in a turbine from 5,000 kPa and 250oC to 500 kPa at the rate of 0.7 kg mol s-1. If the turbine efficiency is 0.80, w hat is the power output of the turbine and what is...
-
The normal freezing point of ethanol (CH3CH2OH) is -117.30 C and its Kfp value is 1.99 C/m. A nonvolatile, nonelectrolyte that dissolves in ethanol is testosterone . How many grams of testosterone,...
-
Farmhill Ltd . had 1 , 5 6 5 , 3 0 0 common shares outstanding on 1 January 2 0 X 6 , the beginning of its 2 0 X 6 fiscal year. During the year, on 1 May, the company issued 5 0 9 , 0 0 0 preferred...
-
Be sure to answer all parts. Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate). Include phase abbreviations: Part 1 out of 2 (a) Provide the...

Study smarter with the SolutionInn App


