(a) With the aid of a phase diagram, explain what is meant by a supercritical fluid. Give...
Question:
(a) With the aid of a phase diagram, explain what is meant by a supercritical fluid. Give examples of commercial processes that involve the use of supercritical fluids.
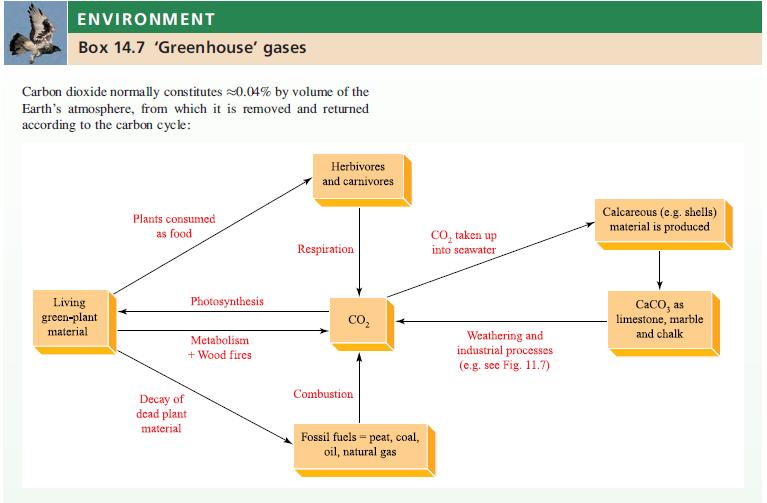
(b) Even though CO2 is classified as a ‘greenhouse gas’ (see Box 14.7), why is the use of supercritical CO2 regarded as being environmentally friendly?

Transcribed Image Text:
ENVIRONMENT Box 14.7 'Greenhouse gases Carbon dioxide normally constitutes 20.04% by volume of the Earth's atmosphere, from which it is removed and returned according to the carbon cycle: Living green-plant material Plants consumed as food Photosynthesis Metabolism + Wood fires Decay of dead plant material Herbivores and carnivores Respiration CO₂ Combustion Fossil fuels peat, coal, oil, natural gas = CO₂ taken up into seawater Weathering and industrial processes (e.g. see Fig. 11.7) Calcareous (e.g. shells) material is produced CaCO, as limestone, marble and chalk
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a Supercritical Fluids A supercritical fluid is a state of matter that exists above its critical temperature and critical pressure The critical temperature Tc is the highest temperature at which the s...View the full answer

Answered By

Alex Maina Muigai
I am a recent graduate of Maseno University, where I earned a Bachelor of Science degree in Chemical Engineering. During my studies, I developed a strong knowledge base in chemical engineering principles, including chemical reaction engineering, fluid mechanics, and thermodynamics. I also gained extensive experience in laboratory techniques and process engineering.
Since graduating, I have been working as a tutor in the field of chemical engineering. I have been providing guidance and support to students in the topics of chemical engineering, including designing chemical processes, designing experiments, and troubleshooting problems. I am also proficient in mathematics, physics, and chemistry, which I use to help my students understand the concepts of chemical engineering.
I have a passion for teaching and helping others understand complex concepts. I have a strong commitment to always providing accurate and reliable information to my students, and I strive to ensure that they have a positive learning experience. I am confident that my knowledge and experience in chemical engineering will be an asset to any student who chooses to work with me.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Explain what is meant by recalculation and reperformance. Give an example of each type of audit evidence. Why are recalculation and reperformance often dual-purpose tests?
-
Explain what is meant by 'a two-bit ADC and draw a diagram to illustrate the outcome of such a ADC being used to digitize a sine wave. Why is it generally desirable to use an ADC with the largest...
-
A health care facility in a metropolitan area is interested in the efficiency of its laboratory turnaround time. Based on data collected over last year, the mean turn around time was found to be 55...
-
Ozarks Camping Equipment, Inc., has established the following direct-material standards for its two products. During May, the company purchased 4,200 yards of tent fabric for its standard model at a...
-
What is your strongest and weakest area of negotiating? How will you improve your negotiating skills? Include two or three of the most important tips you learned that you will use.
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
The director of the MBA program at Salterdine University wants to use DA to determine which applicants to admit to the MBA program. The director believes that an applicants undergraduate grade point...
-
Describe Edgar F. Codd's accomplishments and contributions to the relational model.?
-
Confirm that the structures of [BrF 2 ] + and [BrF 4 ] (9.19 and 9.20) are consistent with the VSEPR model. F Br (9.19) + F J KI Br. F F F (9.20)
-
(a) In aqueous solution at pH 0, Mn 3+ disproportionates to MnO 2 and Mn 2+ . Write equations for the two half-reactions involved in this process. (b) Use Fig. 8.2 to obtain values of E for the...
-
Fill in the blanks with an appropriate word, phrase, or symbol(s). With the negative exponent rule, for x 0, the expression x -5 can be simplified to ________.
-
Doug wants to give Kim a laptop computer that is stored in a locker at the airport. Doug gives Kim the key to the locker and tells her to take the laptop from the locker. Kim says that she doesnt...
-
Ray operates the Apple Spice Restaurant in space that he leases in Village Mall. Village Mall is owned by VM Associates. VM Associates sells the mall to BB Properties. For the rest of the lease term,...
-
Shirley borrows $200,000 from Ridgetop Credit Union to buy a home, which secures the loan. Three years into the term, she stops making payments on it. Ridgetop repossesses and auctions off the...
-
Queenie agrees to rent a bicycle from Ride City Bikes for a day. As a bailor, Ride City must a. deliver the bike to Queenie. b. provide storage of the bike for compensation. c. surrender the bike to...
-
Orley agrees to lease an F-150 Ford truck to Pete, who tells Orley that he plans to use the truck to haul trash and debris from his property. As a bailee, Pete has a responsibility to a. limit his...
-
On February 1, Year 3, Harrier Ltd., a Canadian company, sold goods to a company in a foreign country and took a note receivable for FF6,200,000. The note matures on February 1, Year 5, and bears...
-
Cornell and Roberts are partners who agree to admit Stanley to their partnership. Cornell has a capital balance of $80,000 and Roberts has a capital balance of $120,000. Cornell and Roberts share net...
-
Arrange the acids HSO 4 , H 3 O + , H 4 SiO 4 , CH 3 GeH 3 , NH 3 , HSO 3 F in order of increasing acid strength.
-
With the aid of the table of E and C values (Table 5.5), discuss the relative basicities of (a) Acetone and dimethyl sulfoxide, (b) Dimethyl sulfide and dimethyl sulfoxide. Comment on a possible...
-
Arrange the oxides Al 2 O 3 , B 2 O 3 , BaO, CO 2 , Cl 2 O 7 , SO 3 in order from the most acidic through amphoteric to the most basic.
-
ANSWER THE ONE'S MARKED WRONG: Note: This problem is for the 2022 tax year. Alfred E. Old and Beulah A. Crane, each age 42, married on September 7, 2020. Alfred and Beulah will file a joint return...
-
Common stock value-Variable growth Personal Finance Problem Home Place Hotels, Inc., is entering into a 3-year remodeling and expansion project. The construction will have a limiting effect on...
-
Write Corporate Governance for small business. the board responsibilities and duties and for the directors and all top management?

Study smarter with the SolutionInn App


