Confirm that the structures of [BrF 2 ] + and [BrF 4 ] (9.19 and 9.20)
Question:
Confirm that the structures of [BrF2]+ and [BrF4]− (9.19 and 9.20) are consistent with the VSEPR model.
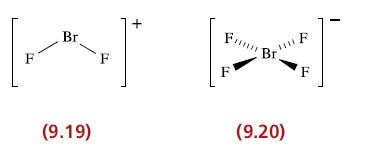
Transcribed Image Text:
F Br (9.19) + F J KI Br. F F F (9.20)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine whether the structures of BrF2 and BrF4 919 and 920 are consistent with the VSEPR Valen...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The analysis described in Problem 1 for the posture measurement data on Shoulder Flexion (SF) may be criticized because information is lost when the 4 observations for a given subject on a given day...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
VSEPR (valence state electron pair repulsion) theory was formulated to anticipate the local geometry about an atom in a molecule (see discussion in Section 25.1). All that is required is the number...
-
The table shows the fees for refund anticipation loans (RALs) offered by an online tax preparation firm. Find the annual rate of interest for each of the following loans. Assume a 360-day year. (A) A...
-
Execucraft, Inc., manufactures upholstered office chairs. The standard cost for material and labor is $ 89.20 per chair. This includes 8 kilograms of direct material at a standard cost of $ 5.00 per...
-
Did the group agree on and were they committed to clear objectives? Explain your answer.
-
Lynn Goldsmith is a photographer known for her photographs of famous musicians. In 1981, Goldsmith had a photography session with the singer Prince. Three years later, Vanity Fair obtained a license...
-
Multiple Choice Questions 1. You are taking a multiple-choice test that awards you one point for a correct answer and penalizes you 0.25 points for an incorrect answer. If you have to make a random...
-
Executives at Southwestern Construction have noticed that the company's construction team in the Phoenix office is more efficient with its resources than the other teams in the Las Vegas, Salt Lake,...
-
Using your answers to problem 7.11, write down expressions for the solubility (in mol dm 3 ) of (a) AgCl, (b) CaCO 3 (c) CaF 2 in terms of K sp . Data from Problem 11 Write down expressions for Ksp...
-
(a) With the aid of a phase diagram, explain what is meant by a supercritical fluid. Give examples of commercial processes that involve the use of supercritical fluids. (b) Even though CO 2 is...
-
On your own or with a partner, choose one of the following consumer products and think about how it could be used as a business product. Then create a business advertisement for your product. a. Pet...
-
Daryll and Sharon Dykes filed a petition for Chapter 7 relief in a federal bankruptcy court, reporting just under $400,000 in assets, over $5.6 million in liabilities, and a monthly income that is...
-
Sue applies for a fire insurance policy for her warehouse from A&I Insurance Company. To obtain a lower premium, she misrepresents the age of the property. The policy is granted. After the warehouse...
-
Ann rents a kayak from Boaters Marina for a days paddling on Clearwater Creek. When Ann takes a break onshore, Donnie steals the kayak. Ann can attempt to a. recover the price of the kayak from...
-
Satellite Communications, Inc., takes out an insurance policy on its plant. For which of the following reasons could the insurer cancel the policy? a. Satellites president appears as a witness in a...
-
Heneli borrows $150,000 from Countywide Credit Union to buy a home. By recording the mortgage, Countywide protects its a. priority against a previously filed lien on the property. b. priority against...
-
On June 1, Year 3, Forever Young Corp. (FYC) ordered merchandise from a supplier in Turkey for Turkish lira (TL) 200,000. The goods were delivered on September 30, with terms requiring cash on...
-
A circular concrete shaft liner with Youngs modulus of 3.4 million psi, Poissons ratio of 0.25, unconfined compressive strength 3,500 psi and tensile strength 350 psi is loaded to the verge of...
-
Write balanced equations for the main reaction occurring when (a) H 3 PO 4 and Na 2 HPO 4 (b) CO 2 and CaCO 3 are mixed in aqueous media.
-
When a pair of aqua cations forms an MOM bridge with the elimination of water, what is the general rule for the change in charge per M atom on the ion?
-
The ions Na + and Ag + have similar radii. Which aqua ion is the stronger acid? Why?
-
Inflation Rate 3 . 0 % Discount Rate ( Nominal ) 1 0 . 0 % Tax Rate 3 0 . 0 % Year Now 2 0 0 1 2 0 0 2 2 0 0 3 New Gross Profit 1 7 . 4 1 2 0 . 5 0 2 1 . 1 1 Old Gross Profit 1 5 . 5 5 1 6 . 0 2 1 6...
-
Essay answer of approximately two-fifty words for each part Part 1 Go online and research various B2B companies. When you find one you would like to examine for this project, begin by taking a look...
-
How do recombinant DNA techniques contribute to the production of biopharmaceuticals, vaccines, and industrial enzymes through microbial fermentation, mammalian cell culture, and transgenic organism...

Study smarter with the SolutionInn App


