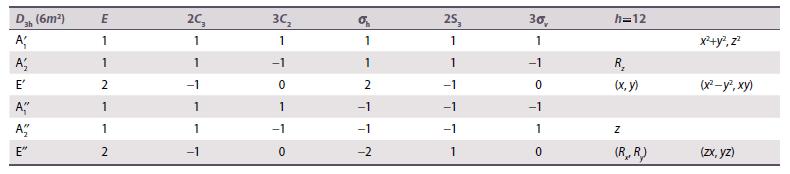
Consider a trigonal prismatic six-coordinate ML 6 complex with D 3h symmetry. Use the D 3h character
Question:
Consider a trigonal prismatic six-coordinate ML6 complex with D3h symmetry. Use the D3h character table (Resources section 4) to divide the d orbitals of the metal into sets of defined symmetry type. Assume that the ligands are at the same angle relative to the xy-plane as in a tetrahedral complex.
Data from Resource section 4.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:





