Consult the Ellingham diagram in Fig. 6.16 and determine if there are any conditions under which aluminium
Question:
Consult the Ellingham diagram in Fig. 6.16 and determine if there are any conditions under which aluminium might be expected to reduce MgO. Comment on these conditions.
Figure 6.16.
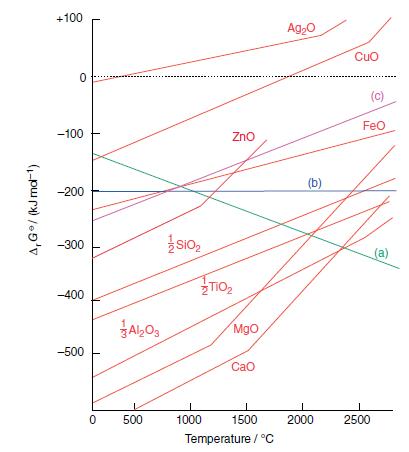
Transcribed Image Text:
A,G*/ (kJ md-¹) +100 0 -100 -200 -300 -400 -500 0 A1203 500 SIO₂ 1000 ZnO TiO₂ Mgo CaO 1500 Temperature / °C Ag₂O (b) 2000 Cuo (c) FeO 2500 (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
In this diagram the temperature range ...View the full answer

Answered By

Joseph Njoroge
I am a professional tutor with more than six years of experience. I have helped thousands of students to achieve their academic goals. My primary objectives as a tutor is to ensure that students do not have problems while tackling their academic problems.
4.90+
10+ Reviews
27+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The public safety department at a large urban university was concerned about criminal activities involving nonstudents stealing bicycles and laptops from students. The campus police designed a study...
-
The IASB was considering additional guidance on reporting revenues when other parties are involved in providing goods and services to a company's customer. The Board has been debating the conditions...
-
Background: A new ownership group has recently purchased ABC Liquors. You have been hired by the new management team to analyze their sales data for the past year and provide them with insights about...
-
An electron experiences the greatest force as it travels 2.9 X 106 m/s in a magnetic field when it is moving north-ward. The force is upward and of magnitude 7.2 X 10-13N. What are the magnitude and...
-
The smart phone market has been dominated by Apple, but recently the Droid has been able to leverage Google's information services into market gains while Blackberry, known for its secure business...
-
Thirty-six percent of U.S. adults favor the use of unmanned drones by police agencies. You randomly select nine U.S. adults. Find the probability that the number of U.S. adults who favor the use of...
-
How do you prevent name clashes when using header files?
-
Gershwin Corporation obtained a franchise from Sonic Hedgehog Inc. for a cash payment of $120,000 on April 1, 2010. The franchise grants Gershwin the right to sell certain products and services for a...
-
XYZ Inc., a well-known manufacturer of inflatable boats is considering a new project, a thickened inflatable boat. Thickened boat is of higher quality and double the price of the normal inflatable...
-
Given the following standard potentials in basic solution and assuming that a reversible reaction can be established on a suitable catalyst, calculate E , r G , and K for the reductions of (a) CrO...
-
In Fig. 6.11, which of the boundaries depend on the choice of Fe 2+ concentration as 10 5 mol dm 3 ? Figure 6.11. +0.8 +0.4 E/V -0.4 -0.8 Fe3+ Fe2+ H,O/H, 024 Fe(OH),(s) Fe(OH), (s) 6 8 10 12 14 pH
-
Consider two mutually exclusive alternatives: If the minimum attractive rate of return is 10%, which alternative should be selected? Year -$100 -$50.0 35 16.5 35 16.5 3 35 16.5 35 16.5 2. 4,
-
Create a new project called04.07Calling Methods in the Mod04Assignments folder. Create a class called MathTrick in the newly created project folder. Complete the static methods in the starter code....
-
A firm operates in a perfectly competitive industry. Suppose it has a short run total cost function given by TC = 1200 + 2Q + 0.03Q2. If the market price is $38, what is the firms profit maximizing...
-
I need help with this coding to figure out why it is not running. public class CommissionEmployee extends Employee { //Declaring instance variables private double grossSales; private double...
-
In this assignment, you will take the category, objects, attributes, and methods that you created in Assignment 5.03 and apply them to the structure of an Object-Oriented Program. Before you begin to...
-
All of the following terms are related to a stock exchange, EXCEPT? Question 5Answer a. Knowledge Process Outsourcing (KPO) b. Net Asset Value (NAV) c. Initial Public Offering (IPO) d. New York Stock...
-
How can you construct a galvanic cell from two substances, each having a negative standard reduction potential?
-
Splitting hairs, if you shine a beam of colored light to a friend above in a high tower, will the color of light your friend receives be the same color you send? Explain.
-
Compound 29.34 reacts with Zn(ClO 4 ) 2 6H 2 O to give a complex [Zn(29.34)(OH)] + that is a model for the active site of carbonic anhydrase. Suggest a structure for this complex. What properties...
-
What is the mitochondrial electron-transfer chain, and what role do quinones play in the chain?
-
Comment on the following observations: (a) Blue copper proteins are not always blue. (b) Two different metalloproteins, both containing [4Fe4S] ferredoxins bound to the protein chain by Cys ligands,...
-
} ) 5. Identify what each of the following methods of the subclass do: override, hide, overload or cause a compiler error. public class ClassA { public void methodOne (int i) () public void...
-
The following sample observations were selected. X 4 5 3 6 10 9 8 Y 4 6 5 7 7 8 3 I. Draw a scatter diagram II. Based on the scatter diagram, does there any appear to be any relationship between X...
-
} 3. Would this program compile/run? If not, why; if yes, what would be the output? public class ConstChain1 { } } public static void main(String[] args) { new SubClass(); System.out.println(); new...

Study smarter with the SolutionInn App


