Figure 5.4 shows the formation of three sp 2 hybrid orbitals (see eqs. 5.35.5). (a) Confirm that
Question:
Figure 5.4 shows the formation of three sp2 hybrid orbitals (see eqs. 5.3–5.5).
(a) Confirm that the directionalities of the three hybrids are as specified in the figure.
(b) Show that eqs. 5.3 and 5.5 correspond to normalized wavefunctions.
Equations
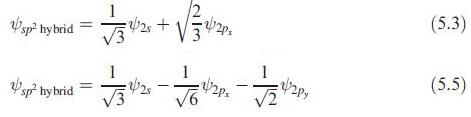
Figure 5.4.
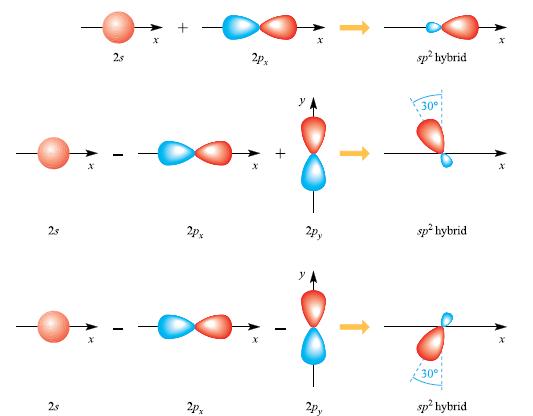
Transcribed Image Text:
sp hybrid 2/25 + 2/20 Psp hybrid 12-12-20 6 2 (5.3) (5.5)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Lets address each part of the question separately a Confirm that the directionalities of the three hybrids are as specified in the figure In Figure 54 ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
DeArmond Corporation has budgeted sales of 54,000 units, target ending finished goods inventory of 9,000 units, and beginning finished goods inventory of 2,700 units. How many units should be...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The bimolecular reaction of chlorine monoxide can result in the formation of three different combinations of products or product channels (rate constant for each reaction is indicated): Determine the...
-
using System; using System.Collections.Generic; using System.ComponentModel; using System.Data; using System.Drawing; using System.Linq; using System.Text; using System.Threading.Tasks; using...
-
Alecs Designs started business in 2012 with total assets of $25,000 and total liabilities of $20,000. At the end of 2012, Alecs Designs total assets were $65,000, and total liabilities were $12,000....
-
Each of the following is intended to be a refutation by logical analogy. Identify the argument being refuted in each and the refuting analogy, and decide whether they do indeed have the same argument...
-
Find the standard deviation of the training times for the (a) male runners. (b) female runners.
-
The graph of the probability distribution of a normally distributed random variable with a mean of 20 and standard deviation of 3 is shown in Figure. The Excel function = NORMINV(Rand( ),20,3) also...
-
Arlington air purification system unadjusted trial balance at December 31,2014
-
(a) The structures of cis- and trans-N 2 F 2 were shown in worked example 3.1. Give an appropriate hybridization scheme for the N atoms in each isomer. (b) What hybridization scheme is appropriate...
-
(a) State what is meant by the hybridization of atomic orbitals. (b) Why does VB theory sometimes use hybrid orbital rather than atomic orbital basis sets? (c) Show that eqs. 5.1 and 5.2 correspond...
-
This problem continues the Canyon Canoe Company situation from Chapter 8. Amber and Zack Wilson are continuing to review business practices. Currently, they are reviewing the companys property,...
-
Identify a weakness of your own that might affect your ability to lead change effectively.
-
What makes an ability (or set of abilities) a core competency? Pick a company you are familiar with (I've picked Apple Company) . Can you identify some of its core competencies What methods do you...
-
What are the key standards and frameworks commonly used by IS auditors during the IS audit process, and how do these standards contribute to the effectiveness and reliability of IS audit activities?...
-
What is the definition of a project risk? What is risk threat and Risk Opportunity? What are the responsibilities of the risk or opportunity owner? What alternatives are there to managing "excessive"...
-
6.1. Determine the transfer function H(s)/Q(s) for the liquid-level system shown in Fig. P61. Resistances R1 and R2 are linear. The flow rate from tank 3 is maintained constant at b by means of a...
-
A researcher took water samples from a stream running through a forest and groundwater in the forest. The samples were collected over the course of a year. Each sample was analyzed for the...
-
On July 1, 2011, Flashlight Corporation sold equipment it had recently purchased to an unaffiliated company for $480,000. The equipment had a book value on Flashlights books of $390,000 and a...
-
The paper Atypical compounds of gases which have been called noble (Chem. Soc. Rev., 2007, 36, 1632) provides a thorough account of the range of compounds formed by Group 18 elements. Among the...
-
Draw the Lewis structures of (a) XeOF 4 , (b) XeO 2 F 2 , (c) XeO 6 2 .
-
Identify the xenon compounds A, B, C, D, and E. D HO C xs F Xe F2 2F E A MeBF B
-
thumbs up if correct A stock paying no dividends is priced at $154. Over the next 3-months you expect the stock torpeither be up 10% or down 10%. The risk-free rate is 1% per annum compounded...
-
Question 17 2 pts Activities between affiliated entities, such as a company and its management, must be disclosed in the financial statements of a corporation as O significant relationships O segment...
-
Marchetti Company, a U.S.-based importer of wines and spirits, placed an order with a French supplier for 1,000 cases of wine at a price of 200 euros per case. The total purchase price is 200,000...

Study smarter with the SolutionInn App


