Four of the lines in the Balmer series are at 656.28, 486.13, 434.05 and 410.17 nm. Show
Question:
Four of the lines in the Balmer series are at 656.28, 486.13, 434.05 and 410.17 nm. Show that these wavelengths are consistent with eq. 1.4.
Data from Equation 1.4.
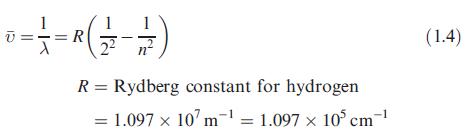
Transcribed Image Text:
1 0 = = = R ( 2²/2 - 1/2 ) R = Rydberg constant for hydrogen = 1.097 x 107 m¹ = 1.097 x 105 cm-¹ 10³ (1.4)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To show that the given wavelengths are consistent with Equation 14 we need to use the Rydberg formul...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
The Bohr model can be applied to singly ionized helium He+ (Z = 2). Using this model, consider the series of lines that is produced when the electron makes a transition from higher energy levels into...
-
A company had a broken printer which they deemed was not worth fixing and thus discarded it. The original cost of the printer was $7,500 and the accumulated depreciation at the time of disposal was...
-
Doreen Dunst started an interior design company called Interiors by Doreen, Inc., on June 1, 2012. The following amounts summarize the financial position of her business on June 14, 2012, after the...
-
Lindsay and her young daughter lived above Rhonda Schmidt, in an apartment complex operated by Towne Properties (TP), where Courtney Haynes, Schmidts boyfriend often stayed, though not on her lease....
-
Outline the general strategy used in metagenomics.
-
Puget Sound Divers is a company that provides diving services such as underwater ship repairs to clients in the Puget Sound area. The companys planning budget for May appears below: Required: During...
-
Capitalized Interest (LIVELY ACE Ch 9 Pt 2) On January 1, Year 1, Romano Tire Service, Inc. signed a contract to have a new service center built for $900,000. On the same day, Romano borrowed...
-
Using data from Appendix 8, construct a graph to show the trend in the third ionization energies of the elements from Li to Kr. Compare the graph with that shown in Fig. 1.16, and rationalize what...
-
Chromium has four isotopes, How many electrons, protons and neutrons does each isotope possess? 50 54 Cr, Cr, Cr and Cr. 53 54 24 24
-
Sketch the polyhedron. Square pyramid.
-
Suppose that Congress passes legislation that establishes a tax credit for small businesses and tax incentives for all businesses that invest in new plant and equipment. a) What is the anticipated...
-
Company K purchased $1 million face value of bonds issued by company J on 1 July 2015 at purchase cost of $1.05 million. The bond had coupon of 6% payable semi-annually and matures on 30 June 2017....
-
If Gemini has a floating rate loan and enters into a pay-fixed-and-receive-floating interest swap transaction with the same principal amount, which of the following statements is the most appropriate...
-
Explain the following terms: (a) Grant date (b) Measurement date (c) Vesting date (d) Vesting conditions (e) Forfeiture rate
-
The consequences of climate change on the economy is a popular topic in the media. Suppose that a series of wildfires destroys crops in the western states at the same time a hurricane destroys...
-
Explain whether a companys suppliers would prefer the company to have a high accounts payable turnover or a low accounts payable turnover.
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
Identify the products from the reaction between the following pairs of reagents. In each case identify the species which are acting as a Lewis acid or a Lewis base in the reactions. (a) CsF + BrF 3...
-
Use the data in Table 5.5 to calculate the enthalpy change for the reaction of iodine with phenol. Table 5.5. TABLE 5.5 Drago-Wayland parameters for some acids and bases* E Acids Antimony...
-
For each of the following processes, identify the acids and bases involved and characterize the process as complex formation or acidbase displacement. Identify the species that exhibit Brnsted...
-
How much heat is required to change one gramme of 0C ice to 100C steam? The latent heat of fusion is 336 J/g and the latent heat of vaporisation is 2268 J/g. The specific heat of water is 4.2 J/(g K).
-
Case 2-Prestige Dance Academy- Page 473 Answer all the case study questions Case 2-Noise Solutions Inc.- Page 467 Answer all the case study questions Case 1- The Ultimate Garage- Page 461 Answer all...
-
The bonds issued by United Corp. bear a coupon of 7 percent, payable semiannually. The bond matures in 20 years and has a $1,000 face value. Currently, the bond sells at $970. The yield to maturity...

Study smarter with the SolutionInn App


