Identify the starting isotopes AE in each of the following syntheses of transactinoid elements: (a) A +
Question:
Identify the starting isotopes A–E in each of the following syntheses of transactinoid elements:
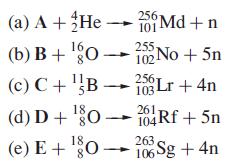
Transcribed Image Text:
(a) A + He → 25 Md+n 16. (b) B+¹0152No +5n (c) C+¹B25Lr + 4n (d) D + ¹80104Rf +5n 18 (e) E+¹80-26Sg +4n 103
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Nuclear Reactions In nuclear physics and nuclear chemistry a nuclear reaction is a process in which ...View the full answer

Answered By

AJIN kuriakose
I have completed B.Tech in Electrical Engineering & Masters in Power & Control From one of the best universities in India. I got the 99.05 percentile in the Gate Electrical Engineering Exam. I can Help students solving assignments in Electrical subjects like Power Electronics, Control system, Analog, Network Theory & Engineering Mathematics. Clear your fundamentals and develop problem-solving skills and analytical skills to crack the exam.
Get guidance and the opportunity to learn from experienced...
I can provide tuition for Electrical engineering subjects (Power Electronics, Digital electronics, Network Theory, Control System & Engineering Mathematics). The toughest subject of Electrical engineering can be made simple in online classes...
I can also solve it.
1 .I can help you with your assignments or exams or quiz or tutoring.
2. Very strict to the deadlines.
Message me for any help in assignments, live sessions. I am here to help students for all assignments, tests and exams and I will make sure you always get _95% In your subject.
Contact me in solution inn for any help in your semester, projects and for many more things . Also feel free to contact me through solution inn and for any advise related to tutoring and how it works here.thank you.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Starting with (S)-2-bromobutane, outline syntheses of each of the following compounds: (a) (b) (c) (d) (R)-CH3CHCH2CH3 OCH2CH3 (R)-CH3CHCH2CH3 CCH3 (R)-CH3CHCH2CH3 SH (R)-CH3CHCH2CH3 SCH3
-
Starting with appropriate unlabeled organic compounds, show syntheses of each of the following: (a) C6H5-C¡C-T (b) (c) CH3CH2CH2OD CH3
-
Starting with ethyne, outline syntheses of each of the following. You may use any other needed reagents, and you need not show the synthesis of compounds prepared in earlier parts of this problem....
-
Aquarius -EAST B Capricornus Horizon Sagittarius SOUTH SUN Scorpius Libra WEST- E Description: If you could see both the Sun and the other stars during the day, this is what the sky would look like...
-
Clean Plate Company produces placemats. Each placemat calls for 0.2 meters of vinyl material; the material should cost $1 per meter. In June, the company manufactured and sold 100,000 placemats....
-
Pharmaceutical companies promote their prescription drugs using television advertising. In a survey of 80 randomly sampled television viewers, 10 indicated that they asked their physician about using...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Apachi Company ended its fiscal year on July 31, 2010. The company??s adjusted trial balance as of the end of its fiscal year is as shown at the top of page 182. Instructions(a) Prepare the closing...
-
1. Convert the following iteration Even method to recursive method: (Ex: EVEN(0,10) Void enas iteration even(int Lint n) { } int i=1: while(i
-
Comment on the observations that the electronic spectra of lanthanoid complexes contain many absorptions some of which are weak and sharp and similar to those of the gas-phase metal ions, and some of...
-
Comment on each of the following observations: (a) H for the formation of [Ln(EDTA)(OH 2 ) x ] (x = 2 or 3) in aqueous solution is nearly constant for all Ln and is almost zero. (b) The value of E...
-
Describe five types of messages.
-
The author's final conclusion is that "increased concentration in food retailing (due to increasing consolidation and foreign investment) has resulted in food prices increases, as retailers get their...
-
Question 1 A basic proposition is that exchange rates adjust so as to maintain the purchasing power parity (PPP): the price of a bundle of goods, expressed in common currency, should be the same...
-
A computer system is required that will support the following small garage business : Customers bring their cars to the garage for servicing and repair. The attendant must check the car in, record...
-
How does the process of endosymbiosis contribute to the evolution of eukaryotic cells, and what evidence supports this theory?
-
find an example problem that can be solved using Machine Learning. In this regards, show how does Machine Learning approach solve that problem. Please thoroughly describes ?!
-
Use Tables VIII, IX, X, and XI of Appendix A to find each of the following F -values: a. F.05, where v1 = 8 and v2 = 5 b. F.01, where v1 = 20 and v2 = 14 c. F.025, where v1 = 10 and v2 = 5 d. F.10,...
-
On April 29, 2015, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon has assets with a fair market value of $1.4 million (basis...
-
Assign a name for each of the following compounds. a. b. c.
-
a. Using the relationships derived in Example Problem 7.1 and the values of the critical constants for water from Table 7.2, calculate values for the van der Waals parameters a, b, and R from z c , T...
-
Assign a name for each of the following compounds. Be sure to assign the configuration of each chirality center and indicate the configuration(s) at the beginning of the name. a. b. c. Me
-
Explain why has cash flow become such an important measure of a firm's financial condition?
-
Describe the key parts of the income statement and distinguish between gross sales and net sales.
-
In 2022, Molly had $2,000 in state income taxes withheld from her wages by her employer. When she files her 2022 taxes, Molly takes the standard deduction and has no taxable income as the result of...

Study smarter with the SolutionInn App


