In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. How might you
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
How might you use 31P NMR spectroscopy to distinguish between Ph2PH and Ph3P?
Table 4.3
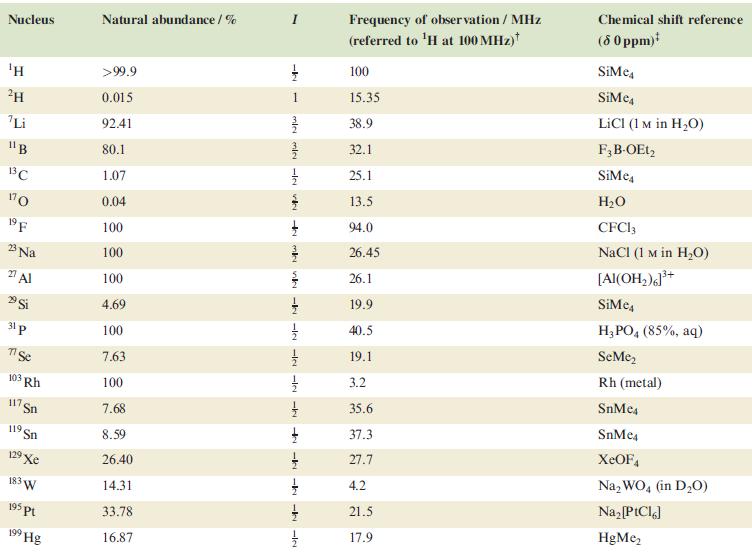
Transcribed Image Text:
Nucleus ΤΗ ²H Li 11B 13C 170 19 F 23 Na 2 Al 29 Si 31 p 7 Se 103 Rh 117. Sn 119 Sn 19 Xe 183 W 195 pt 199 Hg Natural abundance/% >99.9 0.015 92.41 80.1 1.07 0.04 100 100 100 4.69 100 7.63 100 7.68 8.59 26.40 14.31 33.78 16.87 miele v + mk nh -k -le -le-le - -ki -k -k -IN Frequency of observation / MHz (referred to ¹H at 100 MHz) 100 15.35 38.9 32.1 25.1 13.5 94.0 26.45 26.1 19.9 40.5 19.1 3.2 35.6 37.3 27.7 4.2 21.5 17.9 Chemical shift reference (8 0 ppm)* SiMe SiMe4 LICI (1 M in H₂O) F3B-OEt2 SiMe H₂O CFC13 NaCl (1 M in H₂O) [Al(OH₂)]³+ SiMc4 H3PO4 (85%, aq) SeMe₂ Rh (metal) SnMc4 SnMe4 XeOF4 Na₂WO4 (in D₂O) Na₂ [PtCl6] HgMe₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To distinguish between Ph2PH and Ph3P using 31P NMR spectroscopy I would look at the chemical shifts ...View the full answer

Answered By

Huzaifa Salahuddin
Anything that makes my mind focus on thinking attracts me. My major expertise is solving problems of Engineering especially Electrical Engineering and Circuit Analysis. I am also good in problem solving in topics including Oscillation and Waves, Electromagnetism , Light and Optics, Mechanics. I also solve problems in Topics related to Mathematics such as Algebra or Trigonometry. I illustrate each step very clearly so that everyone gets its concepts cleared.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. NaBH 4 contains the tetrahedral [BH4] ion. Although NaBH 4 hydrolyses slowly in water, it is possible to obtain a...
-
How would you use NMR spectroscopy to distinguish between the following pairs of compounds? (a) (b) 'N. 'N- N.
-
How would you use 1 H NMR spectroscopy to distinguish between the following compounds? (a) (b) (c) (d) (e) (f) . . . CI
-
Question 2A) Explain the differences between right issues and bonusissues. Your answer should include the advantages and disadvantagesof both right issues and bonus issues. [ 8marks}b) Explain pos 0...
-
The accounting records for The Skate Shack, Inc., reflected the following amounts at the end of January 2012: Prepare The Skate Shacks multi-step income statement for the fiscal year ended January...
-
A stock is currently priced at $74 and will move up by a factor of 1.20 or down by a factor of 0.80 over the next period. The risk-free rate of interest is 4.2 percent. What is the value of a call...
-
Is there a single standard command-line processor to parse and process argv?
-
Autotech Manufacturing is engaged in the production of replacement parts for automobiles. One plant specializes in the production of two parts: Part 127 and Part 234. Part 127 produced the highest...
-
A car traveling 87.0 km/h is 1500 m behind a truck traveling at 74.0 km/h. How far from its initial position does the car have to travel to catch up to the truck.
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Long-range couplings are often observed between 31 P and 19 F nuclei, between 31 P and 1 H nuclei, but not between...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Why is a coupling constant measured in Hz and is not recorded as a chemical shift difference? Table 4.3 Nucleus H...
-
Hyundai decided to build a new automobile assembly plant in Alabama. a. What factors do you think Hyundai considered in selecting Alabama as the site for the factory? b. Who benefits and who loses...
-
Article-based Discussion Read the article Z Scores A Guide to Failure Prediction, by G.J. Eidleman, The CPA Journal, February 1995. Required: 1. What judgments are involved in the Z-score? 2. What...
-
With the 2-bit predictor, what speedup would be achieved if we could convert half of the branch instructions in a way that replaced each branch instruction with two ALU instructions? Assume that...
-
Bert and Barbara Longfelt support in nursing homes both Berts parents and Barbara's parents. Berts parents are 70 and 68 years of age respectively and have no income except for the $3,600 in Social...
-
Explain the difference between the one-factor and the two-factor ANOVA models.
-
To test whether a bonus plan will improve the monthly sale volume in units, the monthly sale volumes of six salespersons before and after a bonus plan were recorded. At 99 % confidence, determine...
-
Southern Exposure Ltd. begins operations on January 2, 2016. During the year, the following transactions affect shareholders equity: 1. Southern Exposures articles of incorporation authorize the...
-
What are the principal alloying elements in SAE 4340 steel?
-
What is the relation of the possible angular momentum quantum numbers to the principal quantum number?
-
The electron configuration of the transition metals is described by W.H.E. Schwarz in his paper The full story of the electron configurations of the transition elements (J. Chem. Educ., 2010, 87,...
-
Calculate the wavenumber (v=1/) and wavelength of the first transition in the visible region of the atomic spectrum of hydrogen.
-
3. Compare the following alternatives in terms of efficiency and profitability using the B/C and incremental B/C methods. Rate is 4% per year. Benefits Construction $ Life years $/yr A 1,000 400 9 B...
-
Undertake your own research into assistive technologies and provide a one-paragraph overview of: 1. Aided Language Stimulation (ALS). 2.Picture Exchange Communication Systems (PECS). I want big...
-
a. Sold merchandise for cash, $19,530. The cost of the goods sold was $11,720. (Record the sale first.) b. Sold merchandise on account, $8,310. The cost of the goods sold was $4,990. (Record the sale...

Study smarter with the SolutionInn App


