In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Why is a
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
Why is a coupling constant measured in Hz and is not recorded as a chemical shift difference?
Table 4.3
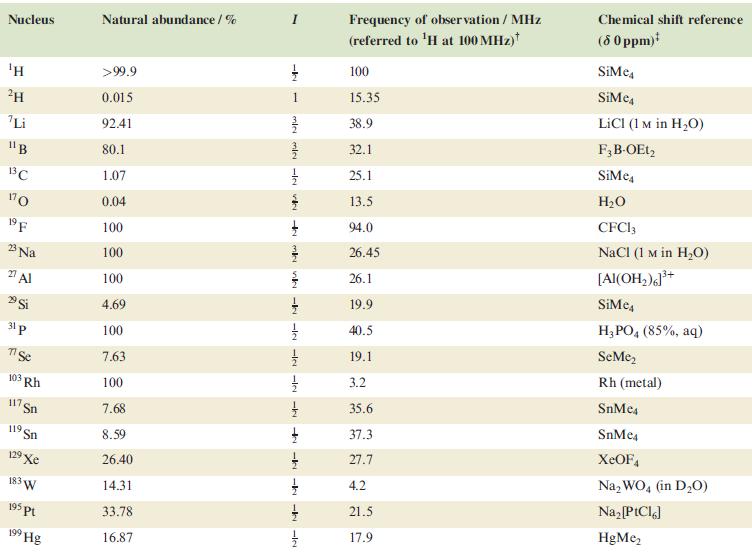
Transcribed Image Text:
Nucleus ΤΗ ²H Li 11B 13C 170 19 F 23 Na 2 Al 29 Si 31 p 7 Se 103 Rh 117. Sn 119 Sn 19 Xe 183 W 195 pt 199 Hg Natural abundance/% >99.9 0.015 92.41 80.1 1.07 0.04 100 100 100 4.69 100 7.63 100 7.68 8.59 26.40 14.31 33.78 16.87 miele v + mk nh -k -le -le-le - -ki -k -k -IN Frequency of observation / MHz (referred to ¹H at 100 MHz) 100 15.35 38.9 32.1 25.1 13.5 94.0 26.45 26.1 19.9 40.5 19.1 3.2 35.6 37.3 27.7 4.2 21.5 17.9 Chemical shift reference (8 0 ppm)* SiMe SiMe4 LICI (1 M in H₂O) F3B-OEt2 SiMe H₂O CFC13 NaCl (1 M in H₂O) [Al(OH₂)]³+ SiMc4 H3PO4 (85%, aq) SeMe₂ Rh (metal) SnMc4 SnMe4 XeOF4 Na₂WO4 (in D₂O) Na₂ [PtCl6] HgMe₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
The coupling constant in nuclear magnetic resonance NMR spectroscopy is measured in Hertz Hz because ...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 19 F NMR spectrum of the octahedral ion [PF5Me] shows two signals ( 45.8 and 57.6 ppm). Why are two signals...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. (a) Predict what you would expect to see in the 15 N NMR spectrum of the isotopically labelled compound cis-[Pt( 15...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Rationalize the fact that the 13 C NMR spectrum of CF 3 CO 2 H consists of two binomial quartets with coupling...
-
A common stock pays an annual dividend that increases by 4% annually and sells for $35 per share. If the market rate of return on this stock is 8%. What is the amount of the ?last dividend paid $1.53...
-
Selected account balances for Megans Mocha at the end of the month are listed below in random order: Accounts...
-
Suppose you buy one SPX put option with a strike of 2100 and write one SPX put option with a strike of 2125. What are the payoffs at maturity to this position for S&P 500 Index levels of 2000, 2050,...
-
What does argv provide to our program?
-
At the end of its first year of operations on December 31, 2012, LAD Company?s accounts show the following. The capital balance represents each partner?s initial capital investment. Therefore, net...
-
3. A 200 kg roller coaster starts from rest at the top of the first hill at a height of 20 m above the ground. The second hill is 15 m above the ground. a. A physics student in line for the ride...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. How might you use 31 P NMR spectroscopy to distinguish between Ph 2 PH and Ph 3 P? Table 4.3 Nucleus H Li 11B 13C...
-
The EI mass spectrum and structure of Cr(CO) 6 is shown in Fig. 4.38. Rationalize the peaks in the spectrum. Why is the EI technique suitable for recording the mass spectrum of Cr(CO) 6 ? Figure 4.38...
-
A trial balance is a formal report. True or false? Please explain.
-
Table 3.1 in the previous chapter presents financial statements for R&E Supplies for the period 2018 through 2021, and Table 3.5 presents a pro forma financial forecast for 2022. Use the information...
-
Professor Preston et al. (1978) studied the effectiveness of bank premiums (stoneware, calculators) given as an inducement to open bank accounts. They randomly selected a sample of 200 accounts each...
-
An SAT review course claims that the variance of test scores of its graduates is less than 150. A random sample of 30 students who took the course is found to have a variance of 225. Assuming a...
-
Bob claims that the variance of the score for the people who took the SAT review course he offers is 100. Fred believes that Bobs students have a variance larger than 100. A random sample of 10 of...
-
A company believed its new toothpaste to have an effect in controlling tooth decay among children. It randomly selected a group of 400 children and gave them the new toothpaste. Another 300 children...
-
DAmbrosio Ltd. has been operating for 10 years. The companys net income and other information since inception are shown below. Required: a. Which year would have been the first year DAmbrosio could...
-
Ask students to outline the reasons why the various elements of culture (social structures and control systems, language and aesthetics, religion and other belief systems, educational systems, etc.)...
-
In the paper Ionization energies of atoms and atomic ions (P.F. Lang and B.C. Smith, J. Chem. Educ., 2003, 80, 938) the authors discuss the apparent irregularities in the first and second ionization...
-
Calculate the wavelength of the line in the atomic spectrum of hydrogen in which n 1 = 1 and n 2 = 3. What is the energy change for this transition?
-
At various times the following two sequences have been proposed for the elements to be included in Group 3: (a) Sc, Y, La, Ac, (b) Sc, Y, Lu, Lr. Because ionic radii strongly influence the chemical...
-
Suppose this information is available for PepsiCo, Inc. for 2023, 2024, and 2025. (in millions) 2023 2024 2025 Beginning inventory $1,926 $2,290 $2,522 Ending inventory 2,290 2,522 2,618 Cost of...
-
K/9 Plus Dog Treats has two departments, Mixing and Packaging. The Packaging Department started in December with work in process inventory of 20,000 pounds (a one-pound bag is a unit of production)....
-
6.4 The real risk-free rate is 2.50%. Inflation is expected to be 2.50% this year and 4.50% during the next 2 years. Assume that the maturity risk premium is zero. What is the yield on 2-year...

Study smarter with the SolutionInn App


