In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Account for the
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
Account for the fact that the 29Si NMR spectrum of a mixture of SiCl4 and SiBr4 that has been standing for 40 h contains five singlets which include those assigned to SiCl4 (δ –19 ppm) and SiBr4 (δ – 90 ppm).
Table 4.3
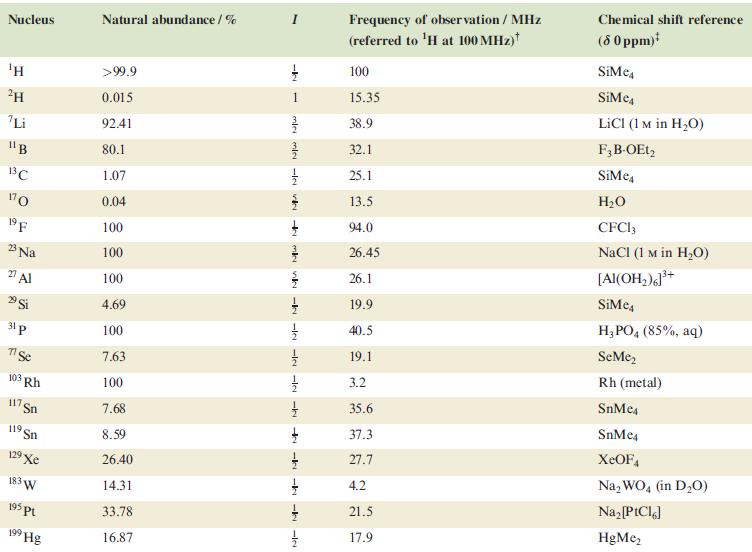
Transcribed Image Text:
Nucleus ΤΗ ²H Li 11B 13C 170 19 F 23 Na 2 Al 29 Si 31 p 7 Se 103 Rh 117. Sn 119 Sn 19 Xe 183 W 195 pt 199 Hg Natural abundance/% >99.9 0.015 92.41 80.1 1.07 0.04 100 100 100 4.69 100 7.63 100 7.68 8.59 26.40 14.31 33.78 16.87 miele v + mk nh -k -le -le-le - -ki -k -k -IN Frequency of observation / MHz (referred to ¹H at 100 MHz) 100 15.35 38.9 32.1 25.1 13.5 94.0 26.45 26.1 19.9 40.5 19.1 3.2 35.6 37.3 27.7 4.2 21.5 17.9 Chemical shift reference (8 0 ppm)* SiMe SiMe4 LICI (1 M in H₂O) F3B-OEt2 SiMe H₂O CFC13 NaCl (1 M in H₂O) [Al(OH₂)]³+ SiMc4 H3PO4 (85%, aq) SeMe₂ Rh (metal) SnMc4 SnMe4 XeOF4 Na₂WO4 (in D₂O) Na₂ [PtCl6] HgMe₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The presence of five singlets in the 29Si NMR spectrum of a mixture of SiCl4 and SiBr4 that has been ...View the full answer

Answered By

Muhammad Rehan
Enjoy testing and can find bugs easily and help improve the product quality.
4.70+
10+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. NaBH 4 contains the tetrahedral [BH4] ion. Although NaBH 4 hydrolyses slowly in water, it is possible to obtain a...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. MeCN solutions of NbCl 5 and HF contain a mixture of octahedral [NbF 6 ] , [NbF 5 Cl] , [NbF 4 Cl 2 ] , [NbF 3 Cl...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Rationalize the fact that at 173 K, 1 H NMR spectroscopy shows that SbMe 5 possesses only one type of Me group....
-
I keep getting the second question wrong. Can you help me to getthat one, please thank you.I tried 8.66, and 8.67 does not are the correct answer A firm has 10 million shares outstanding with a...
-
Mary Rel was hired as the new store manager for the Bargain Bin. The store used four cash registers, and 10 cashiers worked various shifts in the store. Store operations include the following...
-
Jinhee purchased a ticket to a concert to raise money for the local university. The ticket cost $350, but the normal cost of a ticket to this concert is $100. How much is deductible as a charitable...
-
The four alternatives described below are being evaluated: The Incremental IRRs are: a. If the alternatives are independent, which one(s) should be selected if \(M A R R=15.5\) percent/year? b. If...
-
Perform vertical and/or horizontal analysis of the statements and identify two things that appear to be unusual and could be possible symptoms of fraud. The balance sheet and income statement for ABC...
-
One of your clients occasionally smokes cigarettes. They have told you their partner does not know and that they would be very mad with them if they found out. One day their partner asks you if they...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. (a) In the 1 H NMR spectrum of compound 4.21, there is a triplet at 3.60 ppm (J 10.4 Hz). Assign the signal and...
-
Use the VSEPR model to predict the structures of (a) H 2 Se, (b) [BH 4 ] , (c) NF 3 , (d) SbF 5 , (e) [H3O] + , (f) IF 7 , (g) [I 3 ] , (h) [I 3 ] + , (i) SO 3 .
-
Find three online leadership, team roles, or personality assessment tools. Go to testyourself.psychtests.com for examples. How consistent are the results in describing your personality or tendencies?...
-
Yem Company manufactures luggage sets. Yem sells its luggage sets to department stores. Yem expects to sell 2,050 luggage sets for $320 each in January and 2,200 luggage sets for $320 each in...
-
Mark owns a machine shop. In reviewing the shops utility bills for the past 12 months, he found that the highest bill of $2,400 occurred in August when the machines worked 1,000 machine hours. The...
-
Refer to the Packers schedule of cash receipts from customers that you prepared in Short Exercise S22A-17. Now assume that Packerss sales are collected as follows: 50% in the month of the sale 20% in...
-
What is the purpose of variable overhead cost variance?
-
What does profitability mean with regard to performance evaluation in investment centers?
-
On March 1, 2015, Zephur Winds Ltd. purchased a machine for $80,000 by paying $20,000 down and issuing a note for the balance. The machine had an estimated useful life of nine years and an estimated...
-
An access route is being constructed across a field (Figure Q8). Apart from a relatively firm strip of ground alongside the field's longer side AB, the ground is generally marshy. The route can...
-
The article Oxygen, sulfur, selenium, tellurium and polonium by L. Myongwon Lee and I. Vargas-Baca (Annu. Rep. Prog. Chem., Sect. A: Inorg. Chem., 2012, 108, 113) summarizes highlights from the 2011...
-
In their paper Spiral chain O 4 form of dense oxygen (Proc. Natl. Acad. Sci. U.S.A., 2012, 109, 3, 751), L. Zhu and co-workers describe their predicted chain structure for oxygen. Under what...
-
The bond lengths in O 2 , O 2 + , and O 2 2 are 121, 112, and 149 pm, respectively. Describe the bonding in these molecules in terms of molecular orbital theory and use this description to...
-
Find a basis B for the span of the given vectors. 1 2 -1 1 1 0 1 1 0 3 1 1 HONN B =
-
Find the zeros for the polynomial function and give the multiplicity for each zero. State whether the graph crosses the x-axis or touches the x-axis and turns around at each zero. f(x)=8(x+4)(x-7)
-
Complete the square and write the equation in standard form. Then give and radius of the circle. 8x + 8y = 64

Study smarter with the SolutionInn App


