In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Rationalize the fact
Question:
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed.
Rationalize the fact that at 173 K, 1H NMR spectroscopy shows that SbMe5 possesses only one type of Me group.
Table 4.3
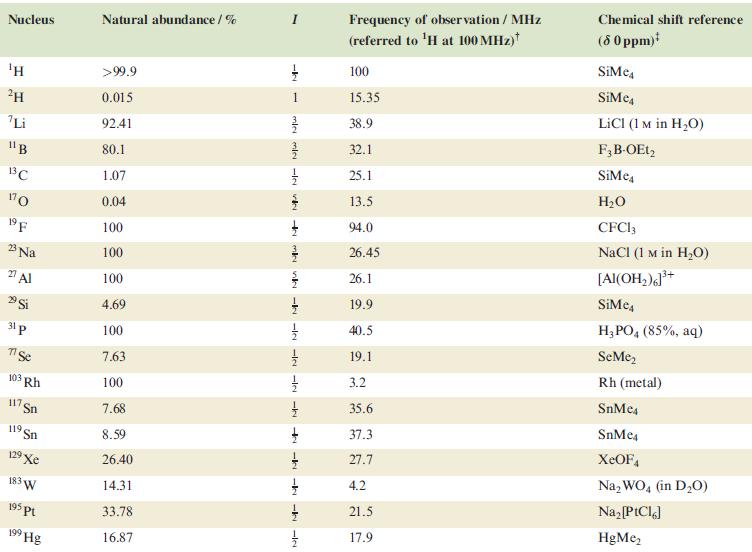
Transcribed Image Text:
Nucleus ΤΗ ²H Li 11B 13C 170 19 F 23 Na 2 Al 29 Si 31 p 7 Se 103 Rh 117. Sn 119 Sn 19 Xe 183 W 195 pt 199 Hg Natural abundance/% >99.9 0.015 92.41 80.1 1.07 0.04 100 100 100 4.69 100 7.63 100 7.68 8.59 26.40 14.31 33.78 16.87 miele v + mk nh -k -le -le-le - -ki -k -k -IN Frequency of observation / MHz (referred to ¹H at 100 MHz) 100 15.35 38.9 32.1 25.1 13.5 94.0 26.45 26.1 19.9 40.5 19.1 3.2 35.6 37.3 27.7 4.2 21.5 17.9 Chemical shift reference (8 0 ppm)* SiMe SiMe4 LICI (1 M in H₂O) F3B-OEt2 SiMe H₂O CFC13 NaCl (1 M in H₂O) [Al(OH₂)]³+ SiMc4 H3PO4 (85%, aq) SeMe₂ Rh (metal) SnMc4 SnMe4 XeOF4 Na₂WO4 (in D₂O) Na₂ [PtCl6] HgMe₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The question at hand refers to the observation in 1H NMR nuclear magnetic resonance spectroscopy tha...View the full answer

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Account for the fact that the 29 Si NMR spectrum of a mixture of SiCl 4 and SiBr 4 that has been standing for 40 h...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. How might you use 31 P NMR spectroscopy to distinguish between Ph 2 PH and Ph 3 P? Table 4.3 Nucleus H Li 11B 13C...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 31 P{ 1 H} NMR spectrum of a CDCl 3 solution of the square planar rhodium(I) complex 4.22 exhibits a doublet of...
-
On January 18, 2021, Marigold Co. paid $450,000 for a three-year insurance policy that covers February 1, 2021 through January 31, 2024. Marigold's year end is June 30, 2021. Assuming she records the...
-
Purpose: To help you understand the importance of cash flows in the operation of a small business. Youre having a tough time figuring out why your actual cash is always coming up less than the amount...
-
Individuals with the genotype bb are 20 percent less fit than individuals with the genotypes BB or Bb. If B mutates to b at a rate of 10 6 per generation, what is the expected frequency of the allele...
-
If a well-behaved investment alternative's internal rate of return (IRR) is equal to MARR, which of the following statements about the other measures of worth for this alternative must be true? 1....
-
Chapman Construction Company is a building contractor specializing in small commercial buildings. The company has the opportunity to accept one of two jobs; it cannot accept both because they must be...
-
At a price of $340, Vurtego sold 1050 pogo sticks per year. After airing on Shark Tank, demand increased, and Vurtego sold 1950 pogo sticks the following year for $460 a unit. Using the mid-point...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. MeCN solutions of NbCl 5 and HF contain a mixture of octahedral [NbF 6 ] , [NbF 5 Cl] , [NbF 4 Cl 2 ] , [NbF 3 Cl...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Explain why the 19 F NMR spectrum of BFCl 2 consists of an approximate 1 :1 :1 :1 quartet. What would you expect to...
-
Repeat Prob. 1114 for a pressure gage reading of 180 kPa. Oil SG = 0.79 Pgage 370 kPa Gasoline SG = 0.70 Pipe 10 cm Mercury Air 45 m 50 cm 22 cm Water SG 13.6
-
What do the profit centers in responsibility reports mean?
-
Calculate the gross profit and operating income for June using absorption costing. Burlington Company reports the following information for June: Sales Revenue Variable Cost of Goods Sold Fixed Cost...
-
Give three benefits of a standard cost system.
-
What is the formula for calculating the predetermined overhead allocation rate?
-
How does indirect labor differ from indirect materials?
-
On January 1, 2016, SugarBear Company acquired equipment costing $150,000, which will be depreciated on the assumption that the equipment will be useful for five years and have a residual value of...
-
In exchange for land, the company received a 12-month note on January 1. The face amount of the note is $1,000, and the stated rate of interest is 13%, compounded annually. The 13% rate is equal to...
-
Tetramethylammonium fluoride (0.70 g) reacts with SF 4 (0.81 g) to form an ionic product. (a) Write a balanced equation for the reaction (b) Sketch the structure of the anion. (c) How many lines...
-
SF 4 reacts with BF 3 to form [SF 3 ][BF 4 ]. Use VSEPR theory to predict the shapes of the cation and anion.
-
Predict whether any of the following will be reduced by thiosulfate ions, SeO 3 2 , in acidic conditions: VO 2+ , Fe 3+ , Cu + , Co 3+ .
-
K A lot is in the shape of a triangle. One side is 400 ft longer than the shortest side, while the third side is 600 ft longer than the shortest side. The perimeter of the lot is 1900 ft. Find the...
-
Financial intermediaries as part of the financial system are very important for a vibrant economy to move funds from surplus units to the deficit units to finance a productive investment. The...
-
A rectangle has an area of 294 cm. Its length and width are whole numbers. (a) What are the possibilities for the two numbers? (b) Which possibility gives the smallest perimeter? (a) In centimeters,...

Study smarter with the SolutionInn App


