Suggest likely products in the following reactions; (the reactions as shown are not necessarily balanced): (a) xLiI
Question:
Suggest likely products in the following reactions; (the reactions as shown are not necessarily balanced):
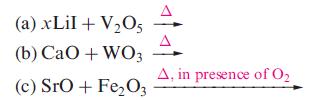
Transcribed Image Text:
(a) xLiI + V₂05 (b) CaO + WO3 (c) SrO + Fe₂O3 A, in presence of 0₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a xLi VO Lithium Vanadate ...View the full answer

Answered By

Ma Kristhia Mae Fuerte
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suggest products for the following reactions (which are not necessarily balanced on the left-hand sides): (a) CsF+ XeF4 (b) SiO + XeOF4 (c) XeF + SbF5 (d) XeF6 + [OH] (e) KrF + HO-
-
Suggest products for the following reactions, which are not necessarily balanced on the left-hand side: (a) KrF + Au (b) XeO3 + RbOH 298 K (c) [XeC1] [SbF1] (d) KrF + B (OTeF5)3 (e) C6F-XeF +...
-
Suggest products of the following reactions, which are not necessarily balanced on the left-hand side: (a) AlMe6+ HO (b) AIR3 + R'NH (c) Me3 SiCl + Na[C5H5] (d) Me SiCl + Li [AIH4]
-
Some food retailers propose subjecting food to a low level of radiation in order to improve safety, but sale of such "irradiated" food is opposed by many people. Suppose a grocer wants to find out...
-
Last year, Panacea Laboratories, Inc., researched and perfected a cure for the common cold. Called Cold-Gone, the product sells for $28.00 per package, each of which contains five tablets. Standard...
-
Convert each temperature as indicated. When appropriate, give your answer to the nearest tenth of a degree. 10F = _________ C.
-
In 2014, political consulting firm Cambridge Analytica developed an app designed to create digital profiles of individuals via their information. Cambridge Analytica collected the data by inviting...
-
At December 31, 2010, Jimenez Company reported the following as plant assets. During 2011, the following selected cash transactions occurred. April 1 Purchased land for $2,130,000. May 1 Sold...
-
Consider the activity-on-arc network diagram below: 1 2 C E 2 4 5 6 3 D 5 5 F G 7 2 8 2 2 1.1 Calculate the early and late event times for each activity in the project network dia- gram. (8) 1.2...
-
(a) MOCVD with Al(O i Pr) 3 as the precursor can be used to deposit -Al 2 O 3 . Outline the principle of MOCVD, commenting on the required properties of the precursors. (b) Fibres of InN can be grown...
-
(a) The structure of YBa 2 Cu 3 O 7 can be described as consisting of rock salt and perovskite layers. Describe the origin of this description. (b) Why is the potential replacement of NbTi by...
-
At the beginning of Year 1, Cactus Company has three employees: A, B, and C. Employee A has three expected years of future service, Employee B has four expected years of future service, and Employee...
-
Amanda, who is in the 32% marginal tax bracket, must decide between two investment opportunities, both of which require an initial cash outlay of $75,000 at the beginning of year 1. This investment...
-
Which is best Network protocol to use for new small organization kind of startup
-
Eagle Enterprises issued 10 year bonds in March 2014 with a face value of $1,000, paying semi-annual coupons with a coupon rate of 6%, and a yield to maturity of 4%. It is now March 2017, what is the...
-
One of the best features of Wireshark is its ability to customize your capture approach to address the problem you are trying to find or solve. Security professionals use Wireshark to research...
-
Give a response justifying why NOT to adjust for inflation on financial statements. What are the positives to not adjusting?
-
To use the t-statistic to test for a difference between the means of two populations, what assumptions must be made about the two populations? About the two samples?
-
Open Text Corporation provides a suite of business information software products. Exhibit 10-9 contains Note 10 from the companys 2013 annual report detailing long-term debt. Required: a. Open Text...
-
Propose a mechanism for the following reaction. NaH Br
-
Acid-catalyzed hydration of 1-methylcyclohexene yields two alcohols. The major product does not undergo oxidation, while the minor product will undergo oxidation. Explane.
-
Calculate S surroundings and S total for part (c) of Problem P5.6. Is the process spontaneous? The state of the surroundings is T = 310.K, P = 0.333 bar.
-
Find the complete solution of the linear system of equations, or show that it is inconsistent. Solve by hand (using substitution or elimination). 6x - 2y = 1 5y-15x = 4
-
Find the equation of the tangent plane to at the point (0, 4, 74). x = z = e+y+y +5
-
Analyse and help me come up with a report on AMC Entertainment holdings' accounting policies Specific Requirements (2 of 5 Slides) Section III. "Accounting Quality Analysis" (Qualitative...

Study smarter with the SolutionInn App


