Suggest products for the following reactions (which are not necessarily balanced on the left-hand sides): (a) CsF+
Question:
Suggest products for the following reactions (which are not necessarily balanced on the left-hand sides):
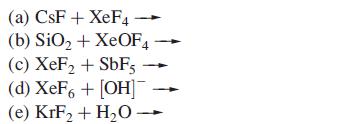
Transcribed Image Text:
(a) CsF+ XeF4 (b) SiO₂ + XeOF4 (c) XeF₂ + SbF5 (d) XeF6 + [OH] (e) KrF₂ + H₂O-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a CSF XeF4 XeF3F CF4 b ...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suggest products for the following reactions, which are not necessarily balanced on the left-hand side: (a) KrF + Au (b) XeO3 + RbOH 298 K (c) [XeC1] [SbF1] (d) KrF + B (OTeF5)3 (e) C6F-XeF +...
-
Suggest products of the following reactions, which are not necessarily balanced on the left-hand side: (a) AlMe6+ HO (b) AIR3 + R'NH (c) Me3 SiCl + Na[C5H5] (d) Me SiCl + Li [AIH4]
-
Suggest products of the following reactions, which are not necessarily balanced on the left-hand side: (a) Mg + C5H6 - (b) MgCl + LiR LiAlH4 (c) RBeCl
-
Lamonda Corp. uses a job order cost system. On April 1, the accounts had the following balances: The following transactions occurred during April: (a) Purchased materials on account at a cost of...
-
Using the information in P23-2B, compute the overhead controllable variance and the overhead volume variance.
-
In problem, use the graph of the function f to solve the inequality. (a) f(x) > 0 (b) f(x) ¤ 0 3 y = 1 3 4 5 x -4 -3 -2 -2 x= 2 X=-1
-
Reconsider Problem 56. Determine which oven should be purchased based on an incremental annual worth analysis. Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The...
-
In Part V of the Pinnacle Manufacturing case, you prepared a performance format audit program. In Part VI, sample sizes will be determined by using nonstatistical or attributes sampling, and the...
-
a.) The following questions refer to the optical cavity in the diagram below. It is excited by a variable frequency source and the detected intensity is displayed on an Optical Spectrum Analyzer....
-
Equation 18.27 showed the preparation of [F 3 AsAuXe][Sb 2 F 11 ] from [F 3 AsAu][SbF 6 ]. Solid [F 3 AsAu][SbF6] contains a distorted [SbF 6 ] ion; one SbF bond is 193 pm, and five are in the range...
-
Comment on the following statements in terms of the properties of the elements mentioned. (a) For many decades, tungsten was used to make filaments in incandescent light bulbs. Tungsten is used in...
-
The planet Mars has two satellites, Phobos and Deimos, in nearly circular orbits. The orbit radii of Phobos and Deimos are 9.38 x 10 3 km and 23.46 x 10 3 km, respectively. The mass of Mars is 6.42 x...
-
A disoriented physics professor drives 3.45 km north, then 2.30 km west, and then 1.65 km south. Part C Find the direction of the resultant displacement measured west of north. Express your answer in...
-
What are the features of the series of -omes? explain
-
What general process is utilized by cells for the intake of liquid and food when the substance is too large to diffuse across the cell membrane?
-
How do allosteric modulators function in the regulation of enzyme activity, and what implications do they hold for drug development targeting enzymatic pathways in complex biological systems?
-
What is the histology section for the circulatory system? explain
-
Roberto has received various gifts over the years. He has decided to dispose of the following assets he received as gifts: a. In 1951, he received land worth $32,000. The donor's adjusted basis was...
-
The Alert Company is a closely held investment-services group that has been very successful over the past five years, consistently providing most members of the top management group with 50% bonuses....
-
In the mass spectrum of bromobenzene (Figure 15.27), the base peak appears at m/z = 77. Figure 15.27 a) Does this fragment contain Br? Explain your reasoning. b) Draw the cationic fragment that...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) OMe . CI H,
-
Butane (C 4 H 10 ) exhibits only two different kinds of protons, shown here in red and blue. (a) Explain why all four protons shown in red are chemically equivalent. (b) Explain why all six protons...
-
An economics professor uses a virtual classroom session and allows students to take the midterm exam as many times as needed until a passing grade is obtained. Following is a record of the number of...
-
2. A key factor in analyzing the industry-level effect of an environmental regulation is to study the so-called market-adjustment (or industry output-adjustment). Suppose that we are looking at the...
-
An analyst is interested in how education affects wage. The analyst specifies the economic model The analyst specifies a statistical model wage Bo+Beduc + e. = wage; Bo+Beduci + ui = unexper; +u. The...

Study smarter with the SolutionInn App


