Suggest products and write balanced equations for each of the following reactions; these are not necessarily balanced
Question:
Suggest products and write balanced equations for each of the following reactions; these are not necessarily balanced on the left-hand side.
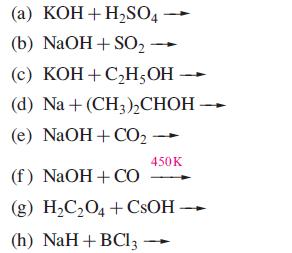
Transcribed Image Text:
(a) KOH + H₂SO4 (b) NaOH + SO₂ (c) KOH + C₂H5OH (d) Na + (CH3)₂CHOH (e) NaOH + CO₂ 450K (f) NaOH + CO (g) H₂C₂O4 +CsOH → (h) NaH+ BC13
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
a KOH H2SO4 Products K2SO4 H2O Balanced equation 2 KOH H2SO4 K2SO4 2 H2O b NaOH SO2 Products Na2SO3 ...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write balanced equations for each of the following reactions (some of these are analogous to reactions shown in the chapter). (a) Aluminum metal reacts with acids to form hydrogen gas. (b) Steam...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Write balanced equations for each of the following processes. a. Chromium-51, which targets the spleen and is used as a tracer in studies of red blood cells, decays by electron capture. b....
-
In baseball, a players batting average is the proportion of times the player gets a hit out of his total number of times at bat. The distribution of batting averages in a recent season for Major...
-
Hiawatha Hydrant Company manufactures fire hydrants in Oswego, New York. The following information pertains to operations during May. Processing time (average per batch)......................4.25...
-
Consider two rectangular surfaces perpendicular to each other with a common edge which is 1.6 m long. The horizontal surface is 0.8 m wide and the vertical surface is 1.2 m high. The horizontal...
-
An asset has the estimated salvage values for various lives, shown in the table below. For each possible life from 1 to 6 by 1, determine the capital recovery cost for MARR of 8 percent/year. EOY NCF...
-
On January 1, 2014, Ven Corporation had the following stockholders equity accounts. Common Stock (no par value, 90,000 shares issued and outstanding) ...$1,600,000 Retained Earnings...
-
Provide three comparisons between hierarchical, network and relational database models. You can present your answer in the table. (b) The relational database model is the most widely used database....
-
Suggest why the solubility of sodium sulfate in water increases to 305K and then decreases.
-
Samples for IR spectroscopy may be prepared as solid state discs by grinding the compound for analysis with an alkali metal halide. Suggest why the IR spectra of K 2 [PtCl 4 ] in KBr and KI discs...
-
The following accounts are in Tan Limiteds December 31, 2023 trial balance: Prepaid Rent $1,600; FV-OCI Investments $62,000; Unearned Revenue $7,000; Land Held for Speculation $119,000; and Goodwill...
-
For a claim of adverse possession to succeed, what four elements must be satisfied?
-
Generally, in a bankruptcy proceeding, any creditors claim is allowed. (True/False)
-
A common carrier is not liable for loss or damage to bailed property in the carriers possession. (True/False)
-
Generally, those who produce personal property have title to it. (True/False)
-
A written agreement is required for all bailments. (True/False)
-
Name the two methods of accounting for contributions, and explain how the methods differ from each other.
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
For which of the following square-planar complexes are isomers possible? Draw all the isomers. [Pt(NH 3 ) 2 (ox)],[PdBrCl(PEt 3 ) 2 ], [Ir(CO)H(PR 3 ) 2 ], [Pd(gly) 2 ].
-
For which of the following tetrahedral complexes are isomers possible? Draw all the isomers. [CoBr 2 Cl 2 ] , [CoBrCl 2 (OH 2 )], [CoBrClI(OH 2 )].
-
The two compounds [RuBr(NH 3 ) 5 ]Cl and [RuCl(NH 3 ) 5 ]Br are what types of isomers?
-
The binomial 2-49 has y - 7 as one of its factors. What is the other factor of the binomial?
-
MAC4865 JANUARY/FEBRUARY 2024 QUESTION 3 (35 marks - 63 minutes) Question 3 consists of 2 independent parts. Part A Ipuleti (Pty) Ltd have two wholly owned subsidiaries, Senoelo (Pty) Ltd and Khaba...
-
Instructions: You are to place the items below in their proper place in the statement of cash flows and also decide whether each item should be a debit or credit Your statement of cash flows will be...

Study smarter with the SolutionInn App


