Suggest products of the following reactions, which are not necessarily balanced on the left-hand side: (a) Mg
Question:
Suggest products of the following reactions, which are not necessarily balanced on the left-hand side:
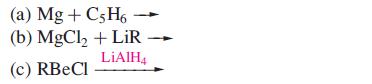
Transcribed Image Text:
(a) Mg + C5H6 - (b) MgCl₂ + LiR LiAlH4 (c) RBeCl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a Mg CsH6 MgH2 Cs Product Magnesium hydride MgH2 and ce...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suggest products for the following reactions (which are not necessarily balanced on the left-hand sides): (a) CsF+ XeF4 (b) SiO + XeOF4 (c) XeF + SbF5 (d) XeF6 + [OH] (e) KrF + HO-
-
Suggest products for the following reactions, which are not necessarily balanced on the left-hand side: (a) KrF + Au (b) XeO3 + RbOH 298 K (c) [XeC1] [SbF1] (d) KrF + B (OTeF5)3 (e) C6F-XeF +...
-
Suggest products of the following reactions, which are not necessarily balanced on the left-hand side: (a) AlMe6+ HO (b) AIR3 + R'NH (c) Me3 SiCl + Na[C5H5] (d) Me SiCl + Li [AIH4]
-
In an effort to check the quality of their cell phones, a manufacturing manager decides to take a random sample of 10 cell phones from yesterdays production run, which produced cell phones with...
-
The comparative balance sheets for Lowery Company show these changes in noncash current asset accounts: accounts receivable decrease $80,000, prepaid expenses increase $28,000, and inventories...
-
Right-circular light passes through a λ/4 retarder whose fast axis is vertical. Describe the emerging polarization state. Did the polarization state shift one quarter of the way around...
-
On April 20, 1992, Daniel Hubbard (plaintiff), a potato farmer, and UTZ Quality Foods, Inc. (UTZ) (defendant), a potato chip manufacturer, entered an installment contract under which Hubbard agreed...
-
The Dubuque Cement Company packs 80-pound bags of concrete mix. Time-study data for the filling activity are shown in the following table. Because of the high physical demands of the job, the...
-
6. A carver begins work on a block of granite that measures 20 cm by 10 cm by 5 cm. Granite has a density of 2.7 g/cm. What is the mass of the piece of granite? a. What do you need to calculate...
-
(a) For the equilibrium comment on the fact that values of K are 1:52 10 8 for R = Me, and 2.3 10 4 for R = Me 2 CHCH 2 . (b) Describe the bonding in Al 2 Me 6 , Al 2 Cl 6 and Al 2 Me 4 (-Cl) 2 ....
-
In 2014, 31 Mt of sulfuric acid was manufactured worldwide. The importance of H 2 SO 4 is reflected in the fact that its consumption in a given country is a direct indicator of that countrys...
-
In problem, use the Rational Zeros Theorem to find all the real zeros of each polynomial function. Use the zeros to factor f over the real numbers. f(x) = x 3 + 2x 2 - 5x 6
-
Consider a sheet of paper 8.2 inches by 11.45 inches. A.) How much force, in newtons, is exerted on one side of the paper by the atmosphere? B.) The mass of a midsized car is about 1000kg. What is...
-
Sheila Rivers is single and lives with her elderly mother, who is infirm. During 2019, Sheila had salary income of $60,000, she contributed $3,500 to a Registered Pension Plan, and she paid union...
-
A shoe collector owned 4 2 pairs of Jordans in 2 0 1 8 . After much research and many online auctions,the shoe collector owned 1 1 2 pairs of Jordans in 2 0 2 2 . a . What is the absolute change in...
-
The duration of the assets is 4, the duration of the liabilities is 5. The size of the assets is 100 and the capital ratio (E/A) is 20%. Calculate the leverage adjusted duration gap.
-
A glass windowpane in a home is 0.62 cm thick and has dimensions of 1.0 m 2.0 m. On a certain day, the indoor temperature is 25 degrees Celsius and the outdoor temperature is 0 degrees Celsius. (a)...
-
The exclusion of state and local bond interest from Federal income tax often is criticized as creating a tax haven for the wealthy. Critics, however, often fail to take into account the effect of...
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
In the first step of the process (oxymercuration), nucleophiles other than water may be used. Predict the product when each of the following nucleophiles is used instead of water: a. b. 1) Hg(OAc)2,...
-
Below are several examples of hydroboration-oxidation. In each case, consider the expected regioselectivity, and then draw the product: a. b. c. 1) , THF 2) H,O2, NaOH 1) BH3 THF 2) H2O2, NaOH
-
Compound A has molecular formula C 5 H 10 . Hydroboration- oxidation of compound A produces 2-methylbutan-1-ol. Draw the structure of compound A: Compound A (C,H10) 1) BH, THF 2) H202, NaOH
-
The amount of the semi-annually payment of a R 65,000 deposited into a sinking fund that earns 6% compounded semb-annually for 45 years is? (Assume payments are made at the end )
-
A bag contains 8 red and 4 yellow marbles. Two marbles are chosen at random and not replaced. What is the probability that 1 marble will be yellow and 1 marble will be red?
-
Frances is parked outside her bank and needs cash today. Her standard checking account balance is sufficient to provide the cash, and she plans to deposit the paycheck she'll receive in a few days to...

Study smarter with the SolutionInn App


