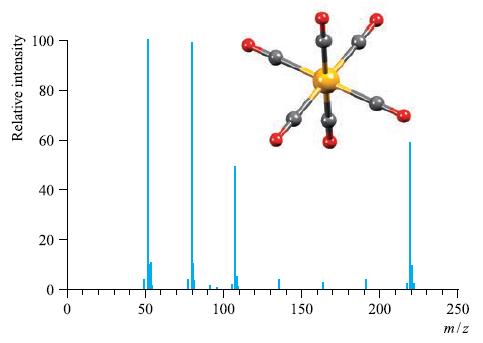
The EI mass spectrum and structure of Cr(CO) 6 is shown in Fig. 4.38. Rationalize the peaks
Question:
The EI mass spectrum and structure of Cr(CO)6 is shown in Fig. 4.38. Rationalize the peaks in the spectrum. Why is the EI technique suitable for recording the mass spectrum of Cr(CO)6?
Figure 4.38
Transcribed Image Text:
Relative intensity 100 80 80 40 20 T O 50 100 150 200 T 250 m/z
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The EI mass spectrum of CrCO6 shows the following peaks A peak at mz 220 which corresponds to the mo...View the full answer

Answered By

Chandrasekhar Karri
I have tutored students in accounting at the high school and college levels. I have developed strong teaching methods, which allow me to effectively explain complex accounting concepts to students. Additionally, I am committed to helping students reach their academic goals and providing them with the necessary tools to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rationalize the indicated fragments in the EI mass spectrum of each of the following molecules by proposing a structure of the fragment and a mechanism by which it is produced. (a)...
-
Suggest a structure for each of the ions corresponding to the following peaks in the EI mass spectrum of ethyl bromide, and give a mechanism for the formation of each ion. (The numbers in parentheses...
-
What type of pump is shown in Fig P11.5? How does it operate?
-
The annual consumption of beef per person was about 64 7 lb in 2000 and about 60 3 lb in 2007 Assume B(t), the annual beef consumption t years after 2000, is decreasing according to the exponential...
-
Suppose Pauls Hardware sells merchandise on account, terms 1/10, n/60, for $700 (cost of the inventory is $380) on March 17, 201 2. Pauls Hardware later received $235 of goods (cost, $105) as sales...
-
A put option with a strike price of $50 sells for $3.20. The option expires in two months and the current stock price is $51. If the risk-free interest rate is 5 percent, what is the price of a call...
-
How many forms of main() are there?
-
Missy Mansion opened a public relations firm called Solid Gold on August 1, 2016. The following amounts summarize her business on August 31, 2016: During September 2016, the business completed the...
-
2. 2. A 20 kg child climbs to the top of a slide that is 3 m above the ground level. She starts from rest and slides down the incline. a. Define and model the energy of the system with Energy Bar...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Why is a coupling constant measured in Hz and is not recorded as a chemical shift difference? Table 4.3 Nucleus H...
-
Both positive and negative-ion ESI mass spectra of [Me 4 Sb][Ph 2 SbCl 4 ] were recorded. In one spectrum, peaks at m/z 181 (100%), 182 (4.5%), 183 (74.6%) and 184 (3.4%) were observed. The other...
-
Ask a student to bring in an ad in which the brand being marketed was at one time a status symbol, but fell out of fashion for a time (e.g., Cadillac, Parker Pen, Izod-Lacoste, etc.). Have the...
-
A manager claims that the standard deviation in their mean delivery time is less than 2.5 days. A sample of 25 customers is taken. The average delivery time in the sample was 4 days with a standard...
-
If the professor decides to reject the null hypothesis (that means passing the student) when the students get eight or more correct answers, what is the probability of a Type I error? Use the...
-
Suppose a political scientist is interested in whether wealth is a determining factor in the individuals propensity to vote. A random sample of 500 people who earned $ 100,000 or more showed that 390...
-
A poll was done to predict the outcome of the upcoming election. Of the 900 potential voters who responded, 500 plan to vote for the incumbent. If a candidate needs 50 % of the votes to win the...
-
A mutual fund manager claims that the returns of stocks in her fund have a variance of no more than .50. A random sample of 25 stocks in her fund has a sample variance of .72. Assuming that the...
-
The following information is available about the capital structure for MacTavish Corporation: Common shares, Class A: 1 million authorized and outstanding. The Class A common shares are entitled to...
-
The graph of an equation is given. (a) Find the intercepts. (b) Indicate whether the graph is symmetric with respect to the x-axis, the y-axis, or the origin. -3 6 -6 3 x
-
When 58.4 nm radiation from a helium discharge lamp is directed on a sample of krypton, electrons are ejected with a velocity of 1.59 10 6 m s 1 . The same radiation ejects electrons from Rb atoms...
-
During 1999 several papers appeared in the scientific literature claiming that d orbitals of Cu 2 O had been observed experimentally. In his paper Have orbitals really been observed? (J. Chem. Educ.,...
-
Show that the following four lines in the Lyman series can be predicted from eqn 1.1: 91.127, 97.202, 102.52, and 121.57 nm. Equation 1.1. 2 R 1 n n (1.1)
-
This is the discussion: 1. Explain when it is more appropriate to use itemized deductions rather than standard deduction? (LO 3) 2. Explain how a tax credit differs from an itemized deduction? (LO...
-
how to construct a syllogism showing that John has parents.
-
Cain Auto Supplies Balance Sheet 31-Dec-23 $20,000 Accounts Payable $76,000 Cash Prepaid Insurance 36,000 Interest Payable 2,000 Accounts Receivable 60,000 Mortgage Payable 216,000 Inventory 204,000...

Study smarter with the SolutionInn App


