The structure of calcite (CaCO 3 ) is shown in Fig. 4.76. Describe how this structure is
Question:
The structure of calcite (CaCO3) is shown in Fig. 4.76. Describe how this structure is related to that of NaCl.
Figure 4.76.
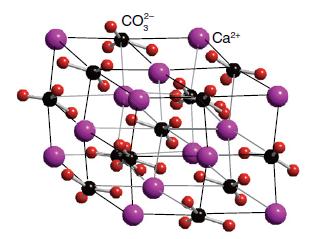
Transcribed Image Text:
CO² Ca²+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The structure of calcite CaCO3 is related to that of NaCl sodium chloride in terms of their overall arrangement of atoms in the crystal lattice Both c...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
What type of pump is shown in Fig P11.5? How does it operate?
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Find H (jw) if its magnitude characteristic is shown in fig 27.
-
According to a survey study conducted by Rossi (2013), the exchange rate predictability depends on: 1 sample period and model 2 the choice of predictor 3 forecast horizon and evaluation method 4 all...
-
What is the value today of $4,000 per year, at a discount rate of 10 percent, if the first payment is received 6 years from today and the last payment is received 20 years from today?
-
Look at Accounts Receivable and Inventory for your company (i.e. the company you work for) or any publicly traded company of your choosing, i.e. Apple (use Yahoo Finance to download their financial...
-
Abbott Industries is a well-known supplier of pharmaceuticals worldwide. Founded by Dr. Wallace Abbott, the company was incorporated in 1900 after he had been developing and making pharmaceuticals...
-
Use the following information to prepare a classified balance sheet for Alpha Co. at the end of 2016. Accounts receivable .....$26,500 Accounts payable ....... 12,200 Cash ............ 20,500 Common...
-
1) (4 marks) Given the function f(x) = x a. Determine the average rate of change on the interval -5 x 2. b. Is it possible to determine the instantaneous rate of change at x = 2? Explain why or why...
-
The perovskite structure, ABX 3 , can be described as a closepacked array of the A and X ions together, with B-type cations in octahedral holes. What proportion of octahedral holes is filled?
-
(a) Calculate the enthalpy of formation of the hypothetical compound KF 2 assuming a CaF 2 structure. Use the BornMayer equation to obtain the lattice enthalpy and estimate the radius of K 2+ by...
-
What is the Sharpe ratio of the best feasible CAL? A pension fund manager is considering three mutual funds. The first is a stock fund, the second is a long-term government and corporate bond fund,...
-
is a general journal entry for a working adjustment the exact opposite of a previously completed adjusting entry?
-
How would you define an effective leader in criminal justice organizations? What are the key characteristics and responsibilities of effective leadership within criminal justice organizations, and...
-
For federal tax purposes, how are the profits of a partnership taxed?
-
In what respect do APT models such as Fama French differ from the Capital Asset Pricing Model ?
-
Cost Value Profit (CVP) analysis is only valid when certain underlying assumptions are met, what are the underlying assumptions of CVP? How might you introduce uncertainty to improve upon the models...
-
It is generally believed that nearsightedness affects about 12% of all children. A school district has registered 170 incoming kindergarten children. a) Can you use the Normal Model to describe the...
-
In a certain school district, 3% of the faculty use none of their sick days in a school year. Find the probability that 5 faculty members selected at random used no sick days in a given year.
-
Write equations for the following processes, involved in the extraction of the elements from their ores: (a) The reduction of boron oxide by Mg; (b) The result of the addition of hot aqueous NaOH to...
-
Comment on each of the following observations. (a) AlF 3 is almost insoluble in anhydrous HF, but dissolves if KF is present. Passage of BF 3 through the resulting solution causes AlF 3 to...
-
(a) Write down, in order, the names and symbols of the elements in group 13; check your answer by reference to the first page of this chapter. (b) Classify the elements in terms of metallic and...
-
10. The probability generating function of the random variable X is given by: Gx(t)= t+ 1+13+ 15 15 15 15 Find: a) The expected value of X. b) E [X(X-1)]. c) The variance.
-
Add or subtract. Simplify by collecting like radical terms if possible. -92+250 -92+250 = (Type an exact answer, using radicals as needed.)
-
Let a = {a1, a2} = {x,1+x} and B = {B1, B2} = {x+x, 2x}. Let J be the transformation from the first question. (i) Write J(a1) as a linear combination of 1 and 2. (ii) Write J(a2) as a linear...

Study smarter with the SolutionInn App


