The structures of R 2 E = ER 2 molecules where E is C, Si, Ge or
Question:
The structures of R2E = ER2 molecules where E is C, Si, Ge or Sn are usually of type A or B shown below:
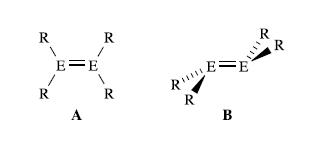
The bonding in the E2-units is described in terms of the interaction of two triplet R2E centres in A, and the interaction of two singlet R2E centres in B. Explain the origins of these descriptions.
Transcribed Image Text:
R R E E A R R REE R B R **** R
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
In the structures of R2E ER2 molecules where E is C Si Ge or Sn the types A and B refer to t...View the full answer

Answered By

Diana Muriuki
As an online math tutor, I have several years of hands-on experience working with students of all ages and skill levels. I hold a Bachelor's degree in Mathematics and a Master's degree in Education. Additionally, I have completed multiple training courses in online teaching and tutoring methods.
Throughout my career, I have worked with students in both individual and group settings, including classroom teaching, after-school tutoring, and online instruction. I am proficient in teaching a wide range of math topics, from basic arithmetic to advanced calculus and statistics.
One of my greatest strengths as a tutor is my ability to adapt my teaching style to meet the unique needs and learning styles of each individual student. I understand that every student is different, and I strive to create a comfortable and supportive learning environment that encourages growth and development.
In addition to my formal education and tutoring experience, I am also a lifelong learner with a passion for mathematics. I am constantly seeking out new resources and methods to improve my own knowledge and skills, and I believe this passion and enthusiasm helps to inspire my students as well.
Overall, my hands-on experience and proficiency as a math tutor are grounded in a combination of formal education, practical experience, and a genuine love of mathematics. I am confident in my ability to help students achieve their goals and succeed in math, and I look forward to the opportunity to work with new students and continue to grow as an educator.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Singlet and triplet carbenes exhibit different properties and show markedly different chemistry. For example, a singlet carbene will add to a cis-disubstituted alkene to produce only...
-
Sodium tripolyphosphate (Na5P3O10) is used in many synthetic detergents. Its major effect is to soften the water by complexing Mg2+ and Ca2+ ions. It also increases the efficiency of surfactants, or...
-
An analysis of comparative balance sheets, the current years income statement, and the general ledger accounts of Judd Corp. uncovered the following items. Assume all items involve cash unless there...
-
What is the easiest way to distinguish between a solution that contains Cu 2+ ions from one that contains Ca 2+ ions?
-
After returning from a skiing vacation in Vermont, Leslie Adel came down with Legionnaires Disease. He claimed it was from the water drunk at the ski resort and provided by Greensprings of Vermont...
-
Martys Barber Shop has one barber. Customers have an arrival rate of 2.2 customers per hour, and haircuts are given with a service rate of 5 per hour. Use the Poisson arrivals and exponential service...
-
Use the attached image and SQL DDL Commands to query the appropriate tables and populate with four rows of data. Please include explanation for script. CUSTOMER PK CustomerID CustomerName Address...
-
(a) Suggest how Na will react with MeC(CH 2 SbCl 2 ) 3 . (b) Comment on aspects of the bonding in the following compound: (c) Cp 2 Ba and (C 5 Me 5 ) 2 Ba both have polymeric structures in the solid...
-
The conversion of (kC 1 -C 5 Me 5 ) 2 SiBr 2 to ( 5 -C 5 Me 5 ) 2 Si is achieved using anthracene/potassium. Outline the role of this reagent.
-
The initial cost of a bridge that is expected to be in place forever is $70 million. Maintenance can be done at 1-, 2-, 3-, or 4-year intervals, but the longer the interval between servicing, the...
-
- 1. For a randomly chosen 50 students from first year students have a mean grade of 75 and standard deviation 10. For an independent sample of 50 students from the second year students have a mean...
-
What does Life After People have to do with the concept of Entropy?
-
Use the formula g'(x) = f'(g(x))" where y = g(x) is the inverse function for y = f(x) to compute g'(2) when f(x) = x3 + x + 2
-
For each of the following matrices A, find eigenvalues and eigenvectors (using Mathe- matica if you like) and draw clear phase plots for the system u' = Au, and classify the origin as an attractor,...
-
Mobile Radio Corp makes car radios, and recently won a contract to install 35,000 radios in a new line of trucks. Without the contract, Mobile's annual cost of goods sold is $3,500,000. The cost of...
-
Ted, an agent for Waxwing Corporation, which is an airline manufacturer, is negotiating a sale with a representative of the U.S. government and with a representative of a developing country. Waxwing...
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
Oxygen reacts with solid glycylglycine C 4 H 8 N 2 O 3 to form urea CH 4 N 2 O, carbon dioxide, and water: 3O 2 (g) + C 4 H 8 N 2 O 3 (s) CH 4 N 2 O(s) + 3CO 2 (g) + 2H 2 O(l) At T = 298 K and 1.00...
-
Identify the reagents that you would use to achieve each of the following transformations: a. b. Br Br
-
Discuss the following statement: Heating an object causes its temperature to increase.
-
How many molecules of O2 are needed to produce eight molecules of H2O through the combustion of C3H8? Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an...
-
8. Using the in problem 7, if 2.50g K_{f}; CuSO 4 are dissolved in 900. mL of 0.30M N*H_{3} what are the concentrations of Cu 2+, N*H_{3}, and Cu(NH 3 ) 4^2+ at equilibrium ?
-
7. The formation constant (K) for Cu(NH 3) 4^2+ is 5 * 10 ^ 13 A) Write the balanced reaction. Cu ^2 + (a 9 ) + 4NH 3 (a 9 ) Cu(NH 3 ) 4 (aq); Kf = [Cv(NH 3) 4 [Cv^ 24 ] Cnv b) Using the reaction and...

Study smarter with the SolutionInn App


