The values of log K for the following two equilibria are 7.23 and 12.27, respectively: Determine (a)
Question:
The values of log K for the following two equilibria are 7.23 and 12.27, respectively:
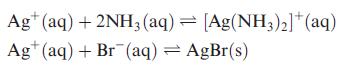
Determine
(a) Ksp for AgBr,
(b) K for the reaction:
![]()
Transcribed Image Text:
Ag+ (aq) + 2NH3(aq) [Ag(NH3)2]+ (aq) Ag*(aq) + Br (aq) = AgBr(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
To determine the values of Ksp for AgBr and K for the given reaction we can use the relationship bet...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Comment on the following observations. (a) In its complexes, Co(III) forms strong bonds to O- and N-donor ligands, moderately strong bonds to P-donor ligands, but only weak bonds to As-donor ligands....
-
The structure of H 5 DTPA (see Box 4.3) is shown below: (a) Write equilibria to show the stepwise acid dissociation of H 5 DTPA. Which step do you expect to have the largest value of K a ? (b) In the...
-
The cable is subjected to a uniform loading of w = 60 kN/m. Determine the maximum and minimum tension in cable. -100 m- 12 m
-
Compare and contrast the three modern theories of international trade.
-
Explain the significance of the Accounting Principles Rule of the AICPA Code of Professional Conduct.
-
A contract is created to refurbish a luxury yacht: new color schemes, new furniture, new wall and floor coverings, new light fixtures, and window treatmentsthe whole works. Of course, it is not just...
-
Waters Hardware Store completed the following merchandising transactions in the month of May. At the beginning of May, Waters ledger showed Cash of $8,000 and Common Stock of $8,000. May 1 Purchased...
-
4. A pharma company you work for makes jars with cream. It claims that the average amount of cream in a jar is at least 90 g. To prove that claim to the customers you decide to test the claim at a...
-
In aqueous solution, boric acid behaves as a weak acid (pK a = 9:1) and the following equilibrium is established: (a) Draw the structures of B(OH) 3 and [B(OH) 4 ]. (b) How would you classify the...
-
The values of pK a (1) and pK a (2) for chromic acid (H 2 CrO 4 ) are 0.74 and 6.49 respectively. (a) Determine values of K a for each dissociation step. (b) Write equations to represent the...
-
What function is used to calculate a loan payment?
-
Financing Decisions and Firm Value What rule should a firm follow when making financing decisions? How can firms create valuable financing opportunities?
-
WACC and Taxes If I use the after-tax cost of debt for my project analysis then I should use the after-tax cost of equity as well. Do you agree with this statement? Explain.
-
Eurasian Natural Resources plc, a Kazakhstani firm listed on the London Stock Exchange, has total non-current assets of $4,938 million. Non-current liabilities are $1,038 million. Current assets,...
-
Preference shares do not offer a corporate tax shield on the dividends paid. Why do we still observe some firms issuing preference shares?
-
Explain what is meant by the cost of equity capital. How is the cost of equity capital linked to the risk of the assets of a firm? How would you use cost of equity in a capital budget analysis?
-
Describe two ratios that measure current liquidity, and compare the information they provide. Which measure is more likely to produce the lowest result for most companies? Why is this the case?
-
Eleni Cabinet Company sold 2,200 cabinets during 2011 at $160 per cabinet. Its beginning inventory on January 1 was 130 cabinets at $56. Purchases made during the year were as follows: February . 225...
-
What are the values of the n, l, and m l quantum numbers that describe the 5f orbitals?
-
The second ionization energies of some Period 4 elements are Identify the orbital from which ionization occurs and account for the trend in values. Ca Sc 1145 1235 Ti 1310 V 1365 Cr 1592 Mn 1509...
-
How many orbitals are there in a shell of principal quantum number n?
-
Sketch in the graph and explain your response. 1. (A) After years of research, cow's milk is believed to be beneficial to bone health. Sketch the supply and demand curves in below to represent the...
-
Why is it important to analyze mixed costs? explain the reason, minimum 200 word.
-
A food service manager is discussing the annual budget with the chief financial officer. The budget includes staffing, food supplies, and two new coolant systems. What is necessary to meet these...

Study smarter with the SolutionInn App


