What is the denticity of the following molecules? Which could act as bridging ligands? Which could act
Question:
What is the denticity of the following molecules? Which could act as bridging ligands? Which could act as chelating ligands?
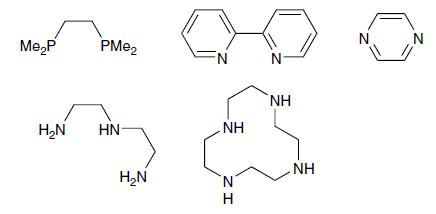
Transcribed Image Text:
Me₂P PMe₂ H₂N HN H₂N FN .ΝΗ ZI N H N NH NH N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
To determine the denticity of the molecules and identify bridging and chelating ligands lets first understand what denticity bridging and chelating ligands mean Denticity Denticity refers to the numbe...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
How many bonds could each of the following chelating ligands form with a metal ion? a. Acetylacetone b. Diethylene triamine c. Salen d. Porphine CH2-C-CH NH2-CH-CH-NH-CH2-CH2-NH2 OH HO NH N N HN
-
Chelating ligands often form more stable complex ions than the corresponding monodentare ligands form with the same donor atoms. For example, where en is ethylenediamine and penten is The increased...
-
The carbonate ion (CO32-) can act as either a monodentate or a bidentate ligand. Draw a picture of CO32- coordinating to a metal ion as a bidentate and as a monodentate ligand. The carbonate ion can...
-
P Corporation acquired an 80% interest in S Corporation two years ago at an implied value equal to the book value of S. On January 2, 2017, S sold equipment with a five-year remaining life to P for a...
-
The following represents the potential outcomes of your first salary negotiation after graduation: Assuming this is a sequential move game with the employer moving first, indicate the most likely...
-
The number of ways 2 captains can be chosen from 28 players on a lacrosse team. Determine whether the situation involves permutations, combinations, or neither. Explain your reasoning.
-
Suppose that in September 2013 a company takes a long position in a contract on May 2014 crude oil futures. It closes out its position in March 2014. The futures price (per barrel) is \($88.30\) when...
-
1. What is the total annual cost of the present ordering policy for part number 650810/ss/R9/o? 2. What would be the lot size for part number 650810/ss/R9/o if FabQual were to use an economic order...
-
Ruler Formula Bar Normal Page Break Page Custom Preview Layout Views Gridlines Headings Zoom 100% Zoom To Selection Window Macros Workbook Views Show Zoom Macros A A1 V A B D E F G H 4 Assume That...
-
Name the octahedral complex ions (a) cis-[CrCl 2 (NH 3 ) 4 ] + , (b) trans-[Cr(NH 3 ) 2 (N-NCS) 4 ] , (c) [Co(C 2 O 4 )(en) 2 ] + .
-
The hydrated chromium chloride that is available commercially has the overall composition CrCl 3 6H 2 O. On boiling a solution, it becomes violet and has a molar electrical conductivity similar to...
-
A dean of a business school wanted to know whether the graduates of her school used a statistical inference technique during their first year of employment after graduation. She surveyed 314...
-
Writ a Java program as follows: Prompt the user which action they want to take: Convert cubic feet to U.S. bushels Convert miles to kilometers Determine graduation with honors title Exit program...
-
4. Apply Euler's method with 4 steps to approximate (3) for the solution of the inital value problem: 21-y 2+y y(1)=0 Present all calculations and display the resulting values in a table. Steph= n ...
-
. Consider the normal-form game pictured here: 2 1 X Y Z A 2,0 1,3 5.x B 5.4 1.3 6,2 All of the payoff numbers are specified, with the exception of that denoted by x. Find a number for x such that...
-
Using C# writeProgram called motelregistration Include classes, objects ,arrays ,loops and exceptions where and if necessary Using c# and fairly simple and organized code the program should prompt a...
-
You plan to purchase a house for $180,000. You have saved $20,000 for your down payment and plan to finance the rest of the cost with a loan which you will you repay in the following way: Equal...
-
What volume of F2 gas, at 25oC and 1.00 atm, is produced when molten KF is electrolyzed by a current of 10.0 A for 2.00 h? What mass of potassium metal is produced? At which electrode does each...
-
Define a traverse in Surveying?
-
Suggest products when Me 3 Sb reacts with the following reagents: (a) B 2 H 6 ; (b) H 2 O 2 ; (c) Br 2 ; (d) Cl 2 followed by treatment with MeLi; (e) MeI; (f) Br 2 followed by treatment with Na[OEt].
-
(a) In what ways do the solid state structures of ( 5 -C 5 R 5 ) 2 Sn for R = H, Me and Ph differ? (b) In the solid state structure of ( 5 -C 5 Me 5 ) 2 Mg, the two cyclopentadienyl rings are...
-
Suggest products when Et 3 SnCl reacts with the following reagents: (a) H 2 O; (b) Na[Cp]; (c) Na 2 S; (d) PhLi; (e) Na.
-
Assume we have a $500,000 mortgage at 3.5% original interest rate, with a 30 year term and monthly payments. The interest rate can be adjusted at the end of each year, and we assume the rate...
-
Translate the following symbolic statements in words. 1)p ~q 2)s~r 3)q ~s Translate the following verbal statements into symbols. 4)Ground moves affect Flying-type Pokmonif some Pokmondo not have a...
-
GloboCorp Industries has hired you to help price a so called "reverse convertible bond" that they are considering issuing as part of a recapitalization. GloboCorp is currently all-equity financed,...

Study smarter with the SolutionInn App


