Which of the following equations show redox reactions: eqs. 15.64, 15.70, 15.73, 15.111 and 15.123? For each
Question:
Which of the following equations show redox reactions: eqs. 15.64, 15.70, 15.73, 15.111 and 15.123? For each redox reaction, indicate which species is being oxidized and which reduced. Confirm that the changes in oxidation states for the oxidation and reduction processes balance.
Equations
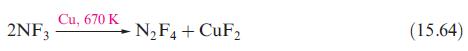
![]()
![]()
![]()
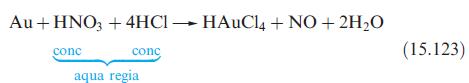
Transcribed Image Text:
2NF3 Cu, 670 K N₂F4 + CuF₂ (15.64)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
Lets go through each of the equations and determine whether they are redox reactions and if so which ...View the full answer

Answered By

Mehwish Aziz
What I have learnt in my 8 years experience of tutoring is that you really need to have a friendly relationship with your students so they can come to you with their queries without any hesitation. I am quite hardworking and I have strong work ethics. Since I had never been one of those who always top in the class and always get A* no matter what, I can understand the fear of failure and can relate with my students at so many levels. I had always been one of those who had to work really hard to get decent grades. I am forever grateful to some of the amazing teachers that I have had who made learning one, and owing to whom I was able to get some extraordinary grades and get into one of the most prestigious universities of the country. Inspired by those same teachers, I am to be like one of them - who never gives up on her students and always believe in them!
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Specify which of the following equations represent oxidation reduction reactions, and indicate the oxidizing agent, the reducing agent, the species being oxidized, and the species being reduced. a....
-
Which of the following are redox reactions? For those that are, indicate which element is oxidized and which is reduced. For those that are not, indicate whether they are precipitation or...
-
Each of the following equations describes a reaction of a compound called methyl formate. To what class of compounds does methyl formate belong? Which reactions require a reducing agent? Which...
-
Suppose the money supply is $250 million dollars and the demand for money is given by Qm D = 400 - 40i, where Qm D is in millions of dollars. A. What is the equilibrium interest rate in this economy?...
-
Explain whether each of the following balance sheet items increases, reduces, or has no direct effect on a companys ability to pay its obligations as they come due. Explain your reasoning. a. Cash....
-
Test the following matrices for singularity. Find the inverses of any that are nonsingular. Multiply the original matrix by its inverse to check your work. Use Mathematica to check your work. a. b. 2...
-
Petitioner Christy Brzonkala met respondents Antonio Morrison and James Crawford at a campus party at Virginia Polytechnic Institute (Virginia Tech), where they were all students. At the party, the...
-
Goldstein Semiconductors experienced the following activity in its Photolithography Department during December. Materials are added at the beginning of the photolithography process. Units: Work in...
-
Recent events have once again shaken and stirred the memory of the nation. Almost immediately after the Civil War, a myth arose painting the conflict with a brush that attempted to blur the lines...
-
(a) Describe the solid state structures of K 3 C 60 and of KC 8 . Comment on any physical or chemical properties of the compounds that are of interest. (b) Comment on the use of lead(II) acetate in a...
-
(a) [SnF 5 ] has a polymeric structure consisting of chains with cis-bridging F atoms. Draw a repeat unit of the polymer. State the coordination environment of each Sn atom, and explain how the...
-
On December 2, 2019, Joan borrowed $2400, agreeing to repay the loan with blended payments of $292 per month, starting on January 2. Interest was charged at 7.8% per annum calculated on the monthly...
-
A view that groups items is very useful when: Select an answer: you do not have permission to create a filtered view. you have a large number of documents in a library. all of these the view cannot...
-
Small cluster size ( ( N ) ) is desirable to Question 3 3 Answer a . . Increase user capacity b . . Reduce CCI c . . Increase traffic intensity d . . Increase SNIR
-
TMP236A2DCKT Temperature sensor, and PIC12F1840-E/P, can they interface with each other? Why?
-
When was Ethereum's genesis block mined? a . . 2 0 1 3 2 0 1 3 b . . 2 0 1 5 2 0 1 5 c . . 2 0 1 8 2 0 1 8 d . . 2 0 0 9 2 0 0 9
-
SharePoint is installed on _ _ _ _ _ . _ _ _ _ _ . Select an answer: a server a local device both of these
-
Enterprise Industries produces Fresh, a brand of liquid laundry detergent. In order to manage its inventory more effectively and make revenue projections, the company would like to better predict...
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
The structure of B 2 O 3 is formed from vertex linked BO 3 planar triangular units while in Al 2 O 3 aluminium ions adopt solely octahedral coordination to oxygen. Explain these observations on the...
-
11 B-NMR is an excellent spectroscopic tool for inferring the structures of boron compounds. With 11 B 11 B coupling ignored, it is possible to determine the number of attached H atoms by the...
-
Starting with B 10 H 14 and other reagents of your choice, give the equations for the synthesis of [Fe(nido-B 9 C 2 H 11 ) 2 ] 2 , and sketch the structure of this species.
-
Revenues FedEx CORPORATION Income Statement Service Revenue (in millions) Interest Revenue Total Revenues Expenses Depreciation Expense 5 Income Tax Expense E Interest Expense Office Expense 58 Rent...
-
Lawn Chopper Company sells two types of lawn mowers. The first one is a basic lawn mower, which has variable costs of $50 and sells for $150. The second type is a riding tractor with variable costs...
-
Executive Desk Company has decided to price its desks at 60% above cost. The problem is they are not sure how to assign factory overhead. Your manager has asked you to price the following two jobs...

Study smarter with the SolutionInn App


