Specify which of the following equations represent oxidation reduction reactions, and indicate the oxidizing agent, the reducing
Question:
Specify which of the following equations represent oxidation– reduction reactions, and indicate the oxidizing agent, the reducing agent, the species being oxidized, and the species being reduced.
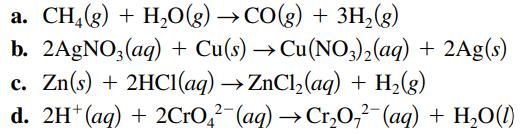
Transcribed Image Text:
a. CH4(g) + H₂O(g) →CO(g) + 3H₂(g) b. 2AgNO3(aq) + Cu(s) → Cu(NO3)₂(aq) + 2Ag(s) c. Zn(s) + 2HCl(aq) → ZnCl₂(aq) + H₂(g) d. 2H+ (aq) + 2CrO²(aq) → Cr₂O₂² (aq) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a This equation represents a combustion reaction which is a type of oxidationreduction ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Which of the following equations represent the correct formula for computing a taxpayer's taxable income Check all that apply. Taxable income = Total income - Exclusions- Adjustments to income...
-
Specify which of the following elements you would expect to have the greatest electron affinity and which would have the least: He, K, Co, S, Cl.
-
Which of the following equations are true for all operators A and B and all functions f and g? (a) (A+ B)f = Af + Bf; (b) A(f + g) = Af + Ag ; (c) (Af)/f = A, provided f 0; (d) AB f = BAf; (e) Af =...
-
You have a net income of $40 000 per year. Your expenses include the following: Rent: $800 per month Insurance: $225 per semi-annually Car Payment: $315 per month Car Expenses: $1 000 per year ...
-
When is the gain on the sale or exchange of securities by a dealer in securities classified as capital gain?
-
Discuss the ethical issues about stress in organizations. Are these real issues to which modern managers should attend? Why or why not?
-
Explain how the notions of effectiveness, efficiency and equity are related to decision-making, sustainable development and accountability.
-
Product S is normally sold for $13 per unit. A special price of $9 is offered for the export market. The variable production cost is $7 per unit. An additional export tariff of 30% of revenue must be...
-
You work for a retail travel consultant. You have been asked to obtain a price for a New Zealand hiking tour on behalf of a customer. The organisation that will deliver the tour services confirms it...
-
Factory work can be boring and monotonous. Employees must work at the pace of the assembly line or machine, with output levels closely prescribed and monitored by management. It is not surprising...
-
Balance the following oxidationreduction reactions that occur in basic solution. a. Cr(s) + CrO2 (aq) Cr(OH)3(s) b. MnO4 (aq) + S(aq) MnS (s) + S(s) c. CN (aq) + MnO4 (aq) CNO (aq) + MnO (s)
-
Balance the following oxidationreduction reactions that occur in acidic solution using the half-reaction method. a. Cu(s) + NO3(aq) Cu+ (aq) + NO(g) b. CrO72 (aq) + Cl(aq) Cr+ (aq) + Cl(g) c. Pb(s)...
-
Solve the system of equations by the addition method. If the system does not have a single ordered pair as a solution, state whether the system is inconsistent or dependent. x + y = 6 -2x + y = -3
-
How can teams focus first and foremost on performance? What are the obstacles to focusing on team performance?
-
What is social chat, and why is it advantageous in an online meeting?
-
Why is it useful to find common ground with others? How do we validate the views and feelings of others?
-
What types of noise are necessary to overcome in a difficult conversation?
-
Why is it useful to commit to listening to peoples stories?
-
During its first year of operations, Fertig Company had credit sales of $3,000,000, of which $400,000 remained uncollected at year-end. The credit manager estimates that $18,000 of these receivables...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
What are the relative values of the chemical shifts observed for nuclei in the spectrometers mentioned in Exercise 15.8a in terms of? (a) Values, (b) Frequencies?
-
The chemical shift of the CH3 protons in diethyl ether is, = 1.16 and that of the CH2 protons is 3.36. What is the difference in local magnetic field between the two regions of the molecule when the...
-
Sketch the appearance of the IH-NMR spectrum of diethyl ether using J = 6.97 Hz and the data in Exercise 15.9b in a spectrometer operating at (a) 350 MHz, (b) 650 MHz.
-
Design a Turing Machine (TM) for the following language, give formal description of the TM and draw state diagram: {w|w is a string over the alphabet {0, 1} and number of 1's in w is divisible by 3}....
-
Jimmy John Hunter owns a small farm outside Winona and was getting very frustrated with turkey vultures that pester his sleeping livestock to the point where his chickens are laying fewer eggs. A few...
-
If the beginning cash balance is $ 5,000, the net cash inflow from operating activities is $20.000, net cash outflow from investing activities is (15,000) and net inflows from financing activities is...

Study smarter with the SolutionInn App


