Which of the following reactions are redox reactions? In those that are, identify the oxidation and reduction
Question:
Which of the following reactions are redox reactions? In those that are, identify the oxidation and reduction processes.
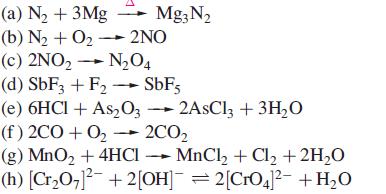
Transcribed Image Text:
(a) N₂ + 3Mg Mg3N₂ (b) N₂ + O₂- 2NO (c) 2NO₂ → N₂04 (d) SbF3 + F₂ → SbF5 (e) 6HCl + As2O32AsCl3 + 3H₂O (f) 2CO+O₂ → 2CO₂ (g) MnO₂ + 4HCI MnCl₂ + Cl₂ + 2H₂O (h) [Cr₂O7]²¯ + 2[OH]¯ = 2[CrO4]²¯ +H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
To determine whether each of the given reactions is a redox reductionoxidation reaction we need to check if there are changes in oxidation states for the elements involved In a redox reaction at least ...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each redox reaction in problem 8.3, confirm that the net increases and decreases in oxidation states balance each other. Data from Problem 8.3 Which of the following reactions are redox reactions?...
-
The commercial production of nitric acid involves the following chemical reactions: (a) Which of these reactions are redox reactions? (b) In each redox reaction identify the element undergoing...
-
Which of the following reactions involves neither oxidation nor reduction? A) N2 + 3H2 ? 2NH3 B) NH4NO2 ? N2 + 2H2O C) Cu + 2Ag+ ? Cu2+ + 2Ag D) 2CrO42- + 2H+ ? Cr2O72- + H2O E) C2H4 + H2 ? C2H6 I...
-
rewrite/downside Integrity and credibility are the ethics of professional practice that Juan Gomez was lacking in this instance. Juan Gomez lacked integrity because he created a conflict of interest...
-
The structure of international trade and tariff systems is highly complex. To maintain order, the United Nations Conference on Trade and Development (UNCTAD) has developed a coding system that...
-
What constitutes internal controls for a payroll department?
-
Defendants Jack and Claire Lein owned and lived on Willow Creek Farm from 1980 through 2004. The farm manager, Stewart, and his girlfriend, plaintiff Tambra Curtis, also lived on the farm during this...
-
The National Oceanic Research Institute is planning a research study on global warming in Antarctica. The 16-month network schedule is presented below. It is followed by budgets for each activity....
-
Find all the complex numbers z such that z += 0. Is the set open or closed? {zC: z*+i=0}
-
What oxidation state change does each metal undergo in the following reactions or half-reactions? (a) [CrO7- + 14H+ +6e2Cr+ (b) 2K + 2HO2KOH + H (c) FeO3 + 2A1 2Fe + AlO3 A (d) [MnO4] + 2HO + 3e MnO...
-
(a) Calculate E Ag + /Ag for a half-cell in which the concentration of silver(I) ions is 0.1moldm 3 (T = 298 K). (b) Are silver(I) ions more or less easily reduced by zinc in this solution than under...
-
Find the volume of the torus generated by revolving the circle (x - 2) 2 + y 2 = 1 about the y-axis.
-
Milton Industries expects free cash flows of $20 million each year. Miltons corporate tax rate is 35%, and its unlevered cost of capital is 9%. The firm also has outstanding debt of $104.13 million,...
-
Colt Systems will have EBIT this coming year of $18 million. It will also spend $7 million on total capital expenditures and increases in net working capital, and have $4 million in depreciation...
-
Consider a simple firm that has the following market-value balance sheet: Next year, there are two possible values for its assets, each equally likely: $1220 and $970. Its debt will be due with 5.1%...
-
Empire Industries forecasts net income this coming year as shown below (in thousands of dollars): Approximately $150,000 of Empires earnings will be needed to make new, positive- NPV investments....
-
NatNah, a builder of acoustic accessories, has no debt and an equity cost of capital of 17%. NatNah decides to increase its leverage to maintain a market debt-to-value ratio of 0.4. Suppose its debt...
-
The MegaMax Theatre is located in the MetroMall and employs six cashiers. Two of them work from 12:30 to 6:00 p.m., and the other four from 6:00 to 11:30 p.m. The cashiers receive payments from...
-
You are interested in investing and are considering a portfolio comprised of the following two stocks. Their estimated returns under varying market conditions are provided: (note: it is difficult to...
-
Describe the delafossite structure type adopted by some compounds of the stoichiometry MMX 2 . Which elemental combinations are known to form this structure type? Discuss their electronic properties...
-
Use ionic radii data (Resource section 1) to suggest possible dopants to increase anion conductivity in (a) PbF 2 (b) Bi 2 O 3 (six-coordinate Bi 3+ ). Resource section 1. Ionic radii are given (in...
-
The compound Fe x O generally has x < 1. Describe the probable metal ion defect that leads to x being less than 1.
-
The primary organizational focus of managerial accounting is on the company's: Select one or more: a. whole b. There is not sufficient information to answer this question. c. Both whole and parts d....
-
Instructions Once logged in, go to My ATI, select the "Learn" tab, and then select the "Nurse Logic 2.0." Watch the Nursing Concepts: Safety Lessons (4:58). Based upon an experience in which you...
-
Carolyn Johnson, CPA, forgot to send account receivable confirmation letters to some of her client's customers during an audit. Such an act is probably an example of Group of answer choices Ordinary...

Study smarter with the SolutionInn App


