What oxidation state change does each metal undergo in the following reactions or half-reactions? (a) [CrO7- +
Question:
What oxidation state change does each metal undergo in the following reactions or half-reactions?
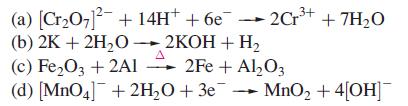
Transcribed Image Text:
(a) [Cr₂O7²- + 14H+ +6e2Cr³+ (b) 2K + 2H₂O2KOH + H₂ (c) Fe₂O3 + 2A1 2Fe + Al₂O3 A (d) [MnO4] + 2H₂O + 3e MnO₂ + 4[OH] -2Cr³+ + 7H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine the oxidation state change for each metal in the given reactions or halfreactions we ne...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Q1. How worried are clients and stakeholders in day-to- day product improvement? 2. the industrial corporation Case for Agility "The struggle is not always to the most powerful, nor the race to the...
-
Question 16 7 pts The First State of Ardmore city reports a net margin of 2.5 percent in its most recent financial report with total interest income of $88 million and total interest expenses of $72...
-
In this exercise, we consider some policy issues related to public support for schoolsand the coalitions between income groups that might form to determine the political equilibrium. A: Throughout,...
-
A spring is at rest in the vertical direction. When a 5 kg mass is placed upon the spring, the length of the spring compresses to 0 . 2 0 meters. The 5 kg mass is removed and replaced by an 8 kg...
-
After watching the video on US-China trade issues, discuss the following: 1. How are consumers affected by the Chinese tire dumping and the subsequent US tariffs? 2. How would you describe the...
-
Connie, a nonexempt employee of Westside Motel, works a standard 6:003:00 p.m. schedule with an hour for lunch. Connie works in a state requiring overtime for hours over 8 per day and for those over...
-
Mrs. Palsgraf was waiting for a train on a platform of a railroad. When a different train came into the station, two men ran to get on that train before it left the station. While one of the men...
-
Kirby Airlines is a small airline that occasionally carries overload shipments for the overnight delivery company Never-Fail, Inc. Never-Fail is a multimillion-dollar company started by Jack Never...
-
For each of the following matrices A Maxn (R), test A for diagonal- izability, and if A is diagonalizable, find an invertible matrix Q and a diagonal matrix D such that Q-1AQ = D. (a) (63) 2 1 3 (b)...
-
(a) Use data from Appendix 11 to construct a potential diagram showing the redox chemistry of vanadium in aqueous solution at pH0. (b) Use your diagram to establish whether any vanadium species is...
-
Which of the following reactions are redox reactions? In those that are, identify the oxidation and reduction processes. (a) N + 3Mg Mg3N (b) N + O- 2NO (c) 2NO N04 (d) SbF3 + F SbF5 (e) 6HCl +...
-
What is the difference between vouching and tracing?
-
CoffeeStop primarily sells coffee. It recently introduced a premium coffee-flavored liquor. Suppose the firm faces a tax rate of 22% and collects the following information. If it plans to finance 15%...
-
Boeing Corporation has just issued a callable (at par) three-year, 4.7% coupon bond with semiannual coupon payments. The bond can be called at par in two years or anytime thereafter on a coupon...
-
Your firm has tangible assets of $103.4 million. You are planning to acquire a firm that is half your firms size. You have bonds with a merger & acquisition covenant that requires the combined firm...
-
Despite the voracious habits of army ants, multiple species of invertebrates have managed to penetrate and exploit their societies. For example, the silverfish Malayatelura ponerophila is an insect...
-
Andyco, Inc., has the following balance sheet and an equity market-to-book ratio of 1.6. Assuming the market value of debt equals its book value, what weights should it use for its WACC calculation?...
-
Explain why inventory is of significance to financial statement users.
-
What is the expected payoff of an investment that yields $5,000 with a probability of 0.15 and $500 with a probability of 0.85? Select one: O a. $325 O b. $5,500 O c. $2,750 O d. $1,175
-
Superconductors are often classified as type I or II. Describe the physical characteristic that determines the classification of a superconductor into one or the other of these types. Classify the...
-
What are the advantages of solgel routes, in comparison with direct high-temperature reaction methods, in the synthesis of complex metal oxides? Why are solgel routes used to prepare finely divided...
-
How might you distinguish experimentally the existence of a solid solution from a series of crystallographic shearplane structures for a material that appears to have variable composition?
-
In a research report, a student is running a two-variable OLS regression to prove that Chinese firms' ROA (a traditional proxy for firm profitability) is adversely affected by CSR spending (measured...
-
Summarize and Discuss how this contents might apply in your personal or professional life. The characteristics of a pure monopoly, the barriers to entry that makeand protect monopolies, price and...
-
1)What kind of benefits giving Asian Infrastructure Investment Bank (AIIB) if Uzbekistan joins in it? 2)What kind of benefits giving EEU if Uzbekistan joins in it? 3)What kind of benefits giving the...

Study smarter with the SolutionInn App


