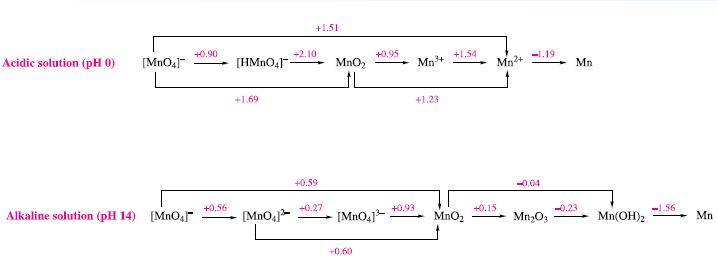
Write balanced half-equations corresponding to the steps shown in the potential diagrams in Fig. 8.2. Figure 8.2
Question:
Write balanced half-equations corresponding to the steps shown in the potential diagrams in Fig. 8.2.
Figure 8.2
Transcribed Image Text:
Acidic solution (pH 0) [MnO4] Alkaline solution (pH 14) [MnO₂] +0.90 +0.56 [HMnO4]- +1.69 [MnO4] +1.51 +2.10 +0.59 +0.27 MnO₂ L +0.95 [MnO4]³ +0,60 +0.93 Mn³+ +1.23 +1.54 MnO₂ Mn²+ +0.15 -1.19 -0.04 Mn₂O3 Mn -0.23 Mn(OH)₂ -1.56 Mn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To write balanced halfequations corresponding to the steps shown in the potential diagrams we need t...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Gruber's (1970) study of n = 104 individuals (discussed in Problem 10), the relationship between blood pressure change (SBPSL) and relative weight (RW), controlling for initial blood pressure...
-
Write equations corresponding to the following energy terms. a. The fourth ionization energy of Se b. The electron affinity of S- c. The electron affinity of Fe3+ d. The ionization energy of Mg e....
-
Write the system of linear equations corresponding to the matrix. 1. 2. 3. 4. -2 -4] -5 3
-
A certain radioactive isotope is a by - product of some nuclear reactors. Due to an explosion, a nuclear reactor experiences a massive leak of this radioactive isotope. Fortunately, the isotope has a...
-
Describe how four strategic goals may affect the decision of where to enter.
-
Rewritethefollowingstatementsfroman"IApproach"toa"YouApproach". 1. Wehavenotreceivedyoursignedinvoicesowecannotprocesspayment. 2. IneedtoknowwhattypeofmodelyouhavebeforeIcandoanything. 3....
-
On April 4, 2014, Athanasios Valsamis lost his appeal to get his money back from a friend to whom he had loaned \($700,000.\) As you will read, this case underscores the consequence of failing to...
-
Jurassic Company owns machinery that cost $900,000 and has accumulated depreciation of $380,000. The expected future net cash flows from the use of the asset are expected to be $500,000. The fair...
-
Flag Faber Manufacturing inc of st paul purchases 9,649 top of the line semiconductor; the maximum backordering quantity in units 502; lead time = 1.5 month ( the firm operates 12 months per year)....
-
Using data from Table 8.1 and from Section 8.3, explain why H 2 is evolved when powdered Ag is heated with a concentrated solution of HI. Data from Table 8.1 Table 8.1 Selected standard reduction...
-
(a) Using appropriate data from Appendix 11, determine E cell for the disproportionation of H 2 O 2 . (b) Calculate G o for this process. (c) Comment on the fact that H 2 O 2 can be stored without...
-
Refer to the information provided in Cornerstone Exercise 10-39. Assume that on July 31, 2007, Titanic discovered that 2006 depreciation was overstated by $50,000. Required: Provide Titanic's...
-
What are some ways to manage a stakeholder relationship closely? Give examples of how you might manage relationships differently based on the unique personalities of different people.
-
Download the template files used in this text from the companion website or from www.kathyschwalbe.com. Review several of the files, and look at examples of how they are used in this text. Also...
-
Discuss some of the examples of poor quality in IT projects presented in the "What Went Wrong?" section. Could most of these problems have been avoided? Why do you think there are so many examples of...
-
Review two different articles about critical chain scheduling. Write a short paper describing how this technique can help improve project schedule management.
-
Read an article or watch a video about a recipient of PMI's Project of the Year award. Search online for information from PMI's website. Write a one-page paper summarizing a winning project, focusing...
-
Lowrate Communications is in the cellular telephone industry. The following selected information has been compiled for the company (in thousands of dollars): Required: a. In each of the three years...
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
Suggest two plausible routes by which a carbonyl ligand in [Mo(Cp)(CO) 3 Me] might exchange for a phosphine. Neither route should invoke the initial dissociation of a CO.
-
(a) What cluster valence electron (CVE) count is characteristic of octahedral and trigonal prismatic complexes? (b) Can these CVE values be derived from the 18-electron rule? (c) Determine the...
-
Ligand substitution reactions on metal clusters are often found to occur by associative mechanisms, and it is postulated that these occur by initial breaking of an MM bond, thereby providing an open...
-
Task 7. Let's get Critical. The figure shows four plants that reproduce without seeds. Look at it and complete the table below. Diagram Plant part used in reproduction. Plant that reproduces this...
-
"It is the great multiplication of the production of all the different arts in consequence of the division of labour, which occasions in a well-governed society, that universal opulence which extends...
-
Mike leases a new pickup by paying $3200 up front and $259 a month over three years. The lease also stipulates he will be charged $0.15 per mile for every mile over 36,000. If he puts 39,928 miles on...

Study smarter with the SolutionInn App


