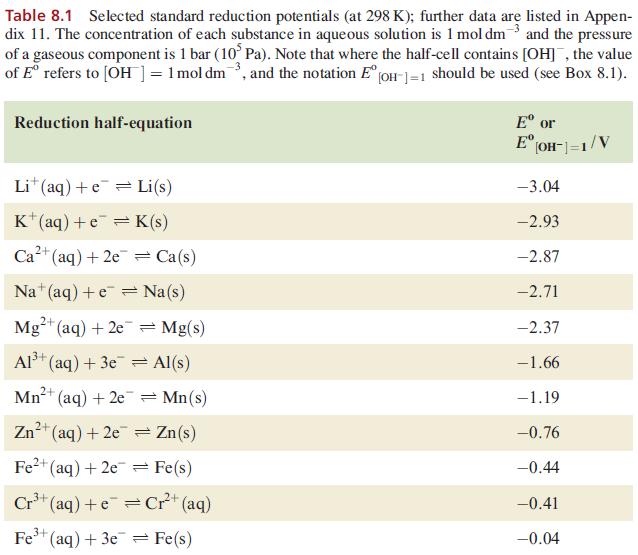
Using data from Table 8.1 and from Section 8.3, explain why H 2 is evolved when powdered
Question:
Using data from Table 8.1 and from Section 8.3, explain why H2 is evolved when powdered Ag is heated with a concentrated solution of HI.
Data from Table 8.1

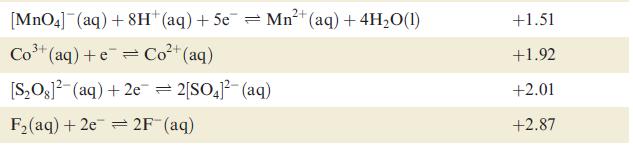
Transcribed Image Text:
Table 8.1 Selected standard reduction potentials (at 298 K); further data are listed in Appen- dix 11. The concentration of each substance in aqueous solution is 1 mol dm3 and the pressure of a gaseous component is 1 bar (10 Pa). Note that where the half-cell contains [OH], the value of Eº refers to [OH] = 1 mol dm³, and the notation Eº [OH-]=1 should be used (see Box 8.1). Reduction half-equation Lit (aq) +eLi(s) K+ (aq) + eK(s) 2+ Ca²+ (aq) +2e= Ca(s) Na (aq) +eNa(s) 2+ Mg²+ (aq) + 2e 3+ Al³+ (aq) + 3e 2+ Mn²+ (aq) + 2e 1²+ (aq) + 2e Fe²+ (aq) + 2e 3+ Cr³+ (aq) + Zn 3+ Fe³+ (aq) + 3e = Mg(s) Al(s) = Mn(s) Zn(s) Fe(s) Cr²+ (aq) = Fe(s) Eº or E [OH-]=1/V -3.04 -2.93 -2.87 -2.71 -2.37 -1.66 -1.19 -0.76 -0.44 -0.41 -0.04
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
To understand why H2 is evolved when powdered Ag silver is heated with a concentrated solution of HI hydrogen iodide we need to consider the redox rea...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following are the financial statements of Swifty Corporation. Swifty Corporation Comparative Balance Sheets December 31 Assets 2019 2018 Cash $37,200 $19,700 Accounts receivable 33,000 18,400...
-
Using data from Table 165 on page 426, assume you purchase a December 1100 (strike price) S&P 500 put option. Compute your total dollar profit or loss if the index has the following values at...
-
Using data from Table 8.4 on bond enthalpies, show that the more C---H bonds a molecule has compared to C-----O and O----H bonds, the more energy it can store.
-
Visit any social media site with news stories that includes a photograph/snapshot of story (mandatory) . Choose ONE news story. Use the questions below to help you determine validity of that ONE news...
-
Summarize the advantages of being a first mover.
-
Based on the following data, would Beth and Roger Simmons receive a refund or owe additional taxes? Adjusted gross income, $42,140 Itemized deductions, $12,240 Credit for child and dependent care...
-
T. Christian Cooper was a partner to Sanders and Richard Campbell d/b/a The Mullen Company. In 2001, Cooper helped bring about a management agreement between The Mullen Co. and Newnan Crossing...
-
Mobile Seating Corporation manufactures seats for automobiles, vans, trucks, and boats. The company has a number of plants, including the Greenville Cover Plant, which makes seat covers. Miriam...
-
On a private flight with capacity for 150 people, $800,000 per person is charged, plus $25,000 for each unsold seat on the plane. How many people must travel on the plane and what will be the price...
-
Determine G o (298 K) for the reaction: given the following data: What does the value of G o tell you about the tendency of precipitated CuCl to disproportionate? 2+ 2CuCl(s) Cu+ (aq) + 2Cl(aq) +...
-
Write balanced half-equations corresponding to the steps shown in the potential diagrams in Fig. 8.2. Figure 8.2 Acidic solution (pH 0) [MnO4] Alkaline solution (pH 14) [MnO] +0.90 +0.56 [HMnO4]-...
-
A customer takes out a loan of $130,000 on January 1, with a maturity date of 36 months, and an annual interest rate of 11%. If 6 months have passed since note establishment, what would be the...
-
Describe three types of software that can assist in project quality management.
-
Discuss the advantages and disadvantages of different ways of distributing project information.
-
How can you use Project 2013 to assist in project human resource management?
-
Discuss the common sources of risk on IT projects and suggestions for managing them. Which suggestions do you find most useful? Which do you feel would not work in your organization? Why?
-
List five reasons why organizations outsource. When should an organization choose not to outsource? Why are some organizations moving their software development work back in-house? Why are some...
-
Big Rock Brewery Inc., which is based in Calgary, Alberta, is a publicly listed company. The companys products are sold across the country in nine provinces and three territories as well as being...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
Lanthanoid coordination compounds rarely exhibit isomerism in solution. Suggest two factors that might cause this phenomenon, explaining your reasoning. (See D. Parker, R.S. Dickins, H. Puschmann, C....
-
Sketch the a 1 symmetry-adapted orbitals for two eclipsed C 5 H 5 ligands stacked together with D 5h symmetry. Identify the s, p, and d orbitals of a metal atom lying between the rings that may have...
-
The compound [Ni( 5 -C 5 H 5 ) 2 ] readily adds one molecule of HF to yield [Ni( 5 -C 5 H 5 )( 4 -C 5 H 6 )] + , whereas [Fe( 5 -C 5 H 5 ) 2 ] reacts with strong acid to yield [Fe( 5 -C 5 H 5 ) 2 H]...
-
Solve the equation by factoring. x=6x+16
-
IV. There are two dates, 1 and 2. There is a market for real bonds at date 1. One unit of bonds pays one unit of goods at date 2 without any risk. 1 The supply of bonds is determined by a simple...
-
Drug Trial: A pharmaceutical company is testing a new drug with 85 patients. They recorded the following frequencies. Drugs Impact on Patient Frequency Strong side effects that resulted in 18...

Study smarter with the SolutionInn App


