A mixture of chloroform and tetrahydrofuran is heated in a closed system at 120 kPa to a
Question:
A mixture of chloroform and tetrahydrofuran is heated in a closed system at 120 kPa to a temperature of 75°C, and two phases are observed to be present. What are the possible compositions of the liquid and vapor phases?
To the Txy diagram for chloroform(1)/tetrahydrofuran(2) at 120 kPa shown in Fig. 12.22.
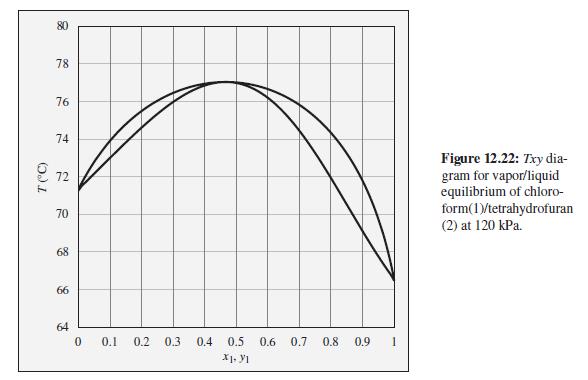
Transcribed Image Text:
78 76 74 Figure 12.22: Txy dia- gram for vapor/liquid equilibrium of chloro- form(1)/tetrahydrofuran (2) at 120 kPa. 72 70 68 66 64 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 X1. Y1 T (C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
At 120 kPa and 75C the given temperature is between the boiling points of chloroform and tetrahyd...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
A chloroform and tetrahydrofuran mixture is heated in a closed system at 120 kPa to a temperature of 70C, and two phases are observed to be present. What are the possible compositions of the liquid...
-
The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 52 kPa. What are the possible compositions of the liquid and vapor phases? To the Pxy diagram for...
-
The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 62 kPa. What are the possible compositions of the liquid and vapor phases? To the Pxy diagram for...
-
Net sales revenue, net income, and common stockholders' equity for Vallen Optical Corporation, a manufacturer of contact lenses, follow for a four-year period. Requirements 1. Compute trend...
-
Although a production possibilities frontier is usually drawn for a country, one could be drawn for the world. Picture the worlds production possibilities frontier. Is the world positioned at a point...
-
The financial statements of The Coca-Cola Company and PepsiCo, Inc. can be accessed at the books website. Instructions Use information found at the books website to answer the following questions....
-
How many years will it take for your savings account to accumulate $\$ 1,000,000$ if it pays $4 \%$ interest per annum compounded semiannually and you deposit $\$ 10,000$ every 6 -months at the end...
-
The top prize for the state lottery is $100,000,000. You have decided it is time for you to take a chance and purchase a ticket. Before you purchase the ticket, you must decide whether to choose the...
-
Problem 3.2A (Static) Using T accounts to record transactions involving assets, liabilities, and owner's equity. LO 3-1, 3-2 The following transactions took place at Willis Counseling Services, a...
-
Fill out the appropriate tax form for Russell and Denise Powell. Tax forms can be obtained from the IRS website. **Be sure you save the form to your computer BEFORE you fill in any information or...
-
Consider a chloroform(1)/tetrahydrofuran(2) mixture with x 1 = 0.80, initially at 70C and 120 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually increased to...
-
Consider a closed vessel initially containing 1 mol of tetrahydrofuran at 50C and 52 kPa. Imagine that pure chloroform is slowly added at constant temperature and pressure until the vessel contains 1...
-
Referring to Example 6.21 (p. 302): a. Calculate P(x < 3|B(25, 3 )). b. How good was the normal approximation? Explain.
-
Why is it important for firms to differentiate themselves from competitors?
-
Complete a competitive analysis grid for Panera Bread.
-
In what ways does a focus on a cost leadership strategy lead to a very different business model than a focus on a differentiation strategy?
-
Describe how the threat of substitute products has the potential to suppress an industrys profitability.
-
What is meant by the term sweat equity?
-
The techniques that are developed for solving linear systems are also applicable to systems with complex coefficients, whose solutions may also be complex. Use Gaussian Elimination to solve the...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
Assuming the validity of R a null's law, do the following calculations for the benzene(1) / toluene(2) system: () Given x1 = 0.33 and T = 100C. find y1 and P, (b) Given y1 = 0.33 and T = HXFC. find...
-
Do alt four pans of Pb.10.9. and compare the results. Discuss any trends that appear.
-
Do alt four pans of Pb.10.9. and compare the results. Discuss any trends that appear.
-
The Casings Plant of Wyoming Machines makes plastics shells for the company's calculators. (Each calculator requires one shell.) For each of the next two years, Wyoming expects to sell 640,000...
-
River Walk Tours is expected to have an EBIT of $184,000 next year. Depreciation, the increase in net working capital, and capital spending are expected to $11,000, $1,500, and $13,000, respectively....
-
Coronado Corporation purchased a depreciable asset for $576000 on January 1, 2023. The estimated salvage value is $54000, and the estimated useful life is 9 years. The straight-line method is used...

Study smarter with the SolutionInn App


