The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 52 kPa.
Question:
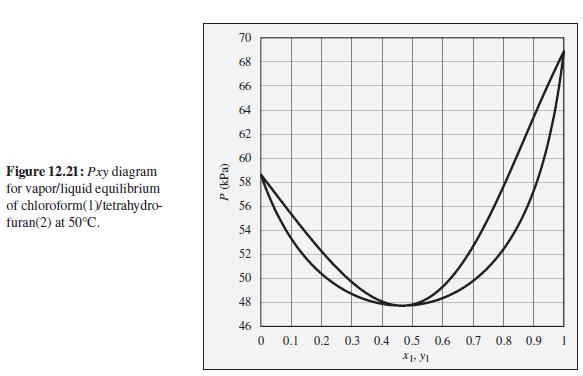
The pressure above a mixture of chloroform and tetrahydrofuran at 50°C is measured to be 52 kPa. What are the possible compositions of the liquid and vapor phases?
To the Pxy diagram for chloroform(1)/tetrahydrofuran(2) at 50°C shown in Fig. 12.21.
Transcribed Image Text:
70 68 66 64 62 60 Figure 12.21: Pxy diagram for vapor/liquid equilibrium of chloroform(1Vtetrahydro- furan(2) at 50°C. 58 56 54 52 50 48 46 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 P (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)

Answered By

Ajeet Singh
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life.
I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge.
I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields.
Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a teacher. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
4.90+
7+ Reviews
15+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
A relativistic rocket is measured to be 50 m long, 2.5 m high, and 2.0 m wide by its pilot. It is traveling at 0.65c (in the direction parallel to its length) relative to an inertial observer. (a)...
-
A mixture of chloroform and tetrahydrofuran is heated in a closed system at 120 kPa to a temperature of 75C, and two phases are observed to be present. What are the possible compositions of the...
-
The pressure of an automobile tire is measured to be 190 kPa (gage) before a trip and 215 kPa (gage) after the trip at a location where the atmospheric pressure is 95 kPa. If the temperature of air...
-
Jay Bhattacharya and Kate Bundorf of Stanford University have found evidence that people who are obese and who work for firms that provide health insurance receive lower wages than workers at those...
-
1. Define consumers surplus. 2. Define producers surplus. 3. Explain how trade is most commonly restricted. 4. Explain how tariffs affect consumers and producers surplus. 5. Explain how quotas affect...
-
A stock is expected to pay a dividend of $0.75 at the end of the year. The required rate of return is rs = 12.5%, and the expected constant growth rate is g = 8.5%. What is the current stock price?...
-
You have just borrowed $\$ 10,000$ and will be required to make monthly payments of $\$ 227.53$ for the next five years in order to fully repay the loan. What is the implicit interest rate on this...
-
The balance sheet items for The Oven Bakery (arranged in alphabetical order) were as follows at August 1, 2011. (You are to compute the missing figure for Retained Earnings.) During the next two...
-
Green Trifle Bakery discovered some errors for 2020 and 2021. What would be the impact on net income for each year if the errors were left undetected? Error Ending inventory in 2020 2020 2021 was...
-
In the United Kingdom, the currency drain ratio is 0.38 of deposits and the reserve ratio is 0.002. In Australia, the quantity of money is $150 billion, the currency drain ratio is 33 percent of...
-
Consider a binary (two-species) system in vapor/liquid equilibrium. Enumerate all of the combinations of intensive variables that could be fixed to fully specify the intensive state of the system.
-
The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 62 kPa. What are the possible compositions of the liquid and vapor phases? To the Pxy diagram for...
-
Indicate whether each statement is true or false. (a) The sequence of amino acids in a protein, from the amine end to the acid end, is called the primary structure of the protein. (b) Alpha helix and...
-
Which computer-based research system contains the text of tax treaties?
-
Where can the historical record of a court case be found?
-
Who may practice before the Internal Revenue Service?
-
Find the value of z|2 for the following values of a. .01 b. c. a = .002 = (a) a (b) = .03 (c) a = .002
-
Assume that Nathan County has levied its current year taxes and all revenue recognition criteria for property taxes have been met. The amount levied was $775,000, of which 2% is deemed to be...
-
Prove directly that the matrix Does not haven an LU factorization. A =
-
Show that if A is any m n matrix, then Im A = A and AIn = A.
-
(a) What is the enthalpy change needed to change 3 kg of liquid water at 0C to steam at 0.1 MPa and 150C? (b) What is the enthalpy change needed to heat 3 kg of water from 0.4 MPa and 0C to steam at...
-
The energy balance can be developed for just about any process. Since our goal is to learn how to develop model equations as well as to simply apply them, it is valuable practice to obtain the...
-
One mole of an ideal gas (C p = 5R/2) in a closed piston/cylinder is expanded from T i = 500 K, P i = 0.6 MPa to P f = 0.1 MPa by the following pathways. For each pathway, calculate U, H, Q, and W EC...
-
Let's assume your lab balloons, when filled with air, each had a mass of 3.00 grams. In a variation of your lab activity, you attach one of these balloons to a string such that the distance from the...
-
Samantha normally requires 1 3 7 0 0 kJ ( about 3 2 7 4 Calories ) of food energy per day. If Samantha consumes 1 4 3 8 5 kJ per day, she will steadily gain weight. How much time must Samantha spend...
-
Five resistors with known resistances R = 5.00 2, R2 = 5.00 2, R3 = 3.00 Q2, R4 = 5.00 Q, and R5 = 5.00 Q are connected to a battery with emf = 7.00 V as shown in the figure. Please answer in Sl...

Study smarter with the SolutionInn App


